

林业科学 ›› 2021, Vol. 57 ›› Issue (9): 130-139.doi: 10.11707/j.1001-7488.20210913
宋玄1,2,3,王泽华1,虞国跃1,王凡1,4,单双2,张永军2,王山宁1,*
收稿日期:2019-11-26
出版日期:2021-09-25
发布日期:2021-11-29
通讯作者:
王山宁
基金资助:Xuan Song1,2,3,Zehua Wang1,Guoyue Yu1,Fan Wang1,4,Shuang Shan2,Yongjun Zhang2,Shanning Wang1,*
Received:2019-11-26
Online:2021-09-25
Published:2021-11-29
Contact:
Shanning Wang
摘要:
目的: 研究白蜡窄吉丁气味结合蛋白AplaOBP2在触角中的表达定位,解析AplaOBP2重组蛋白的配体结合特性及配体活性,旨在通过气味结合蛋白筛选白蜡窄吉丁新的信息化合物。方法: 原核表达白蜡窄吉丁气味结合蛋白AplaOBP2,采用免疫组织化学技术研究AplaOBP2在白蜡窄吉丁触角中的表达定位,通过荧光竞争结合试验分析AplaOBP2重组蛋白与58种化合物的结合特性,并进一步通过触角电位仪和"Y"型嗅觉仪测定AplaOBP2的配体对白蜡窄吉丁成虫的活性。结果: 在原核表达系统中成功表达AplaOBP2重组蛋白。免疫定位结果显示AplaOBP2在触角嗅觉感器——锥形感器Ⅰ的淋巴液表达。荧光竞争结合试验结果表明,AplaOBP2蛋白能够与反-2-己烯醛、反-2-庚烯醛、苯甲醛、4'-乙基苯乙酮、3',4'-二甲氧基苯乙酮和β-紫罗兰酮6种配体结合,其解离常数KD值分别为4.44,4.17,5.20,2.91,3.45和0.63 μmol·L-1。白蜡窄吉丁雌雄成虫对10 mg·mL-1的6种配体均有触角电位反应。行为学试验表明,在10 mg·mL-1刺激剂量条件下,反-2-己烯醛对雌成虫有显著引诱作用,β-紫罗兰酮对雌虫表现出明显的趋避作用。结论: 白蜡窄吉丁气味结合蛋白AplaOBP2在嗅觉感器表达,能够选择性结合醛类和酮类物质,推测其在嗅觉识别中发挥功能。气味结合蛋白可作为靶蛋白,鉴定对白蜡窄吉丁具有吸引或趋避活性的信息化合物。
中图分类号:
宋玄,王泽华,虞国跃,王凡,单双,张永军,王山宁. 白蜡窄吉丁气味结合蛋白AplaOBP2的定位、配体结合特性及配体活性[J]. 林业科学, 2021, 57(9): 130-139.
Xuan Song,Zehua Wang,Guoyue Yu,Fan Wang,Shuang Shan,Yongjun Zhang,Shanning Wang. Localization, Ligand Binding Characteristics and Ligand Activity of an Odorant Binding Protein, AplaOBP2, from Agrilus planipennis[J]. Scientia Silvae Sinicae, 2021, 57(9): 130-139.
表1
重组AplaOBP2蛋白与候选配体结合能力①"
| 配体 Ligand | 来源 Source | CAS号 CAS No. | 纯度 Purity(%) | IC50/(μmol·L-1) | KD/(μmol·L-1) |
| 醇类Alcohols | |||||
| 1-己醇1-hexanol* | TCI | 111-27-3 | >98.0 | — | — |
| 反-2-己烯-1-醇trans-2-hexen-1-ol | TCI | 928-95-0 | >95.0 | — | — |
| 顺-2-己烯-1-醇cis-2-hexen-1-ol | TCI | 928-94-9 | >93.0 | — | — |
| 反-3-己烯-1-醇trans-3-hexen-1-ol | Sigma | 928-97-2 | 97.0 | — | — |
| 顺-3-己烯-1-醇cis-3-hexen-1-ol* | TCI | 928-96-1 | >97.0 | — | — |
| 反4-己烯-1-醇trans-4-hexen-1-ol | Sigma | 928-92-7 | ≥96.0 | — | — |
| 1-辛醇1-octanol | TCI | 111-87-5 | >99.0 | — | — |
| 2-苯乙醇2-phenylethyl alcohol | TCI | 60-12-8 | >98.0 | — | — |
| 醛类Aldehydes | |||||
| 丁醛Butyraldehyde | TCI | 123-72-8 | >98.0 | — | — |
| 戊醛Valeraldehyde | TCI | 110-62-3 | >95.0 | — | — |
| 己醛Hexanal* | TCI | 66-25-1 | >98.0 | — | — |
| 反-2-己烯醛trans-2-hexenal* | TCI | 6728-26-3 | >97.0 | 7.41±0.20 | 4.44±0.12 |
| 反-2-庚烯醛trans-2-heptenal | TCI | 18829-55-5 | >95.0 | 6.95±0.10 | 4.17±0.06 |
| 正辛醛n-octanal | TCI | 124-13-0 | >98.0 | — | — |
| 壬醛Nonanal* | TCI | 124-19-6 | >95.0 | — | — |
| 苯甲醛Benzaldehyde | TCI | 100-52-7 | >98.0 | 8.66±0.31 | 5.20±0.18 |
| 烷类Alkanes | |||||
| 正辛烷Octane | TCI | 111-65-9 | >97.0 | — | — |
| 癸烷Decane* | TCI | 124-18-5 | >99.0 | — | — |
| 十一烷Undecane | TCI | 1120-21-4 | >99.0 | — | — |
| 十二烷Dodecane* | TCI | 112-40-3 | >99.0 | — | — |
| 十三烷Tridecane | TCI | 629-50-5 | >99.0 | — | — |
| 酯类Esters | |||||
| 苯乙酸甲酯Methyl phenylacetate | TCI | 101-41-7 | >99.0 | — | — |
| 水杨酸甲酯Methyl salicylate* | TCI | 119-36-8 | >99.0 | — | — |
| 丁酸顺式-3-己烯酯cis-3-hexenyl butyrate* | Sigma | 16491-36-4 | ≥98.0 | — | — |
| 苯甲酸甲酯Methyl benzoate | TCI | 93-58-3 | >99.0 | — | — |
| 丙烯酸异丁酯Isobutyl acrylate | TCI | 106-63-8 | >99.0 | — | — |
| 丁酸己酯Hexyl butyrate | TCI | 2639-63-6 | >98.0 | — | — |
| 乙酸反-2-己烯酯trans-2-hexenyl acetate | TCI | 2497-18-9 | >97.0 | — | — |
| 丁酸反-2-己烯基酯trans-2-hexenyl butyrate | TCI | 53398-83-7 | >93.0 | — | — |
| 乙酸己酯Hexyl acetate* | TCI | 142-92-7 | >99.0 | — | — |
| 乙酸顺-3-己烯酯cis-3-hexenyl acetate* | TCI | 3681-71-8 | >97.0 | — | — |
| 惕各酸异丁酯Isobutyl tiglate | TCI | 61692-84-0 | >96.0 | — | — |
| 酮类Ketones | |||||
| 2-庚酮2-heptanone | TCI | 110-43-0 | >98.0 | — | — |
| 2-辛酮2-octanone | TCI | 111-13-7 | >98.0 | — | — |
| 4′-乙基苯乙酮4′-ethylacetophenone* | TCI | 937-30-4 | >97.0 | 4.85±0.14 | 2.91±0.09 |
| 3′, 4′-二甲氧基苯乙酮 3′, 4′-dimethoxyacetophenone | TCI | 1131-62-0 | >98.0 | 5.76±0.17 | 3.45±0.10 |
| β-紫罗兰酮β-ionone | TCI | 14901-07-6 | >95.0 | 1.05±0.05 | 0.63±0.03 |
| 萜烯类Terpenes | |||||
| β-蒎烯β-pinene* | 麦克林Maclin | 127-91-3 | ≥95 | — | — |
| (1R)-(+)-α-蒎烯(1R)-(+)-α-pinene* | TCI | 7785-70-8 | >97.0 | — | — |
| 月桂烯Myrcene* | 麦克林Maclin | 123-35-3 | ≥90.0 | — | — |
| 3-蒈烯3-carene* | 麦克林Maclin | 13466-78-9 | 90.0 | — | — |
| (+)-柠檬烯(+)-limonene* | TCI | 5989-27-5 | >95.0 | — | — |
| 松油醇Terpineol | Sigma | 8000-41-7 | ≥96 | — | — |
| α-松油烯α-terpinene | TCI | 99-86-5 | >90.0 | — | — |
| 橙花叔醇Nerolidol* | TCI | 7212-44-4 | >97.0 | — | — |
| α-葎草烯α-humulene* | TCI | 6753-98-6 | >93.0 | — | — |
| α-可巴烯α-copaene* | 化学慧Chemhui | 3856-25-5 | >90.0 | — | — |
| 香叶醇Geraniol | TCI | 106-24-1 | >96.0 | — | — |
| 金合欢烯Farnesene* | Sigma | 502-61-4 | >90.0 | — | — |
| 罗勒烯Ocimene* | Sigma | 13877-91-3 | ≥90.0 | — | — |
| β-石竹烯β-caryophyllene* | TCI | 87-44-5 | >90.0 | — | — |
| (-)-氧化石竹烯(-)-caryophyllene oxide* | Sigma | 1139-30-6 | 95.0 | — | — |
| 芳樟醇Linalool* | TCI | 78-70-6 | >96.0 | — | — |
| 1, 8-桉树脑1, 8-cineole* | TCI | 470-82-6 | >99.0 | — | — |
| 柠檬醛Citral | TCI | 5392-40-5 | >96.0 | — | — |
| (-)-香茅醛(-)-citronellal | TCI | 5949-05-3 | >96.0 | — | — |
| 其他Others | |||||
| 四氢吡咯Pyrrolidine | TCI | 123-75-1 | >98.0 | — | — |
| 吲哚Indole* | TCI | 120-72-9 | >99.0 | — | — |
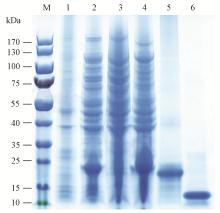
图1
白蜡窄吉丁AplaOBP2重组蛋白SDS-PAGE分析 M: 蛋白质分子质量标准Protein molecular weight marker; 1: 未诱导的大肠杆菌菌体Non-induced Escherichia coli; 2: 诱导的大肠杆菌菌体Induced E. coli; 3: 上清液Supernatant; 4: 包涵体蛋白Inclusion body protein; 5: 带His标签AplaOBP2蛋白Recombinant AplaOBP2 with His-tag; 6: 无His标签AplaOBP2蛋白Recombinant AplaOBP2 without His-tag."
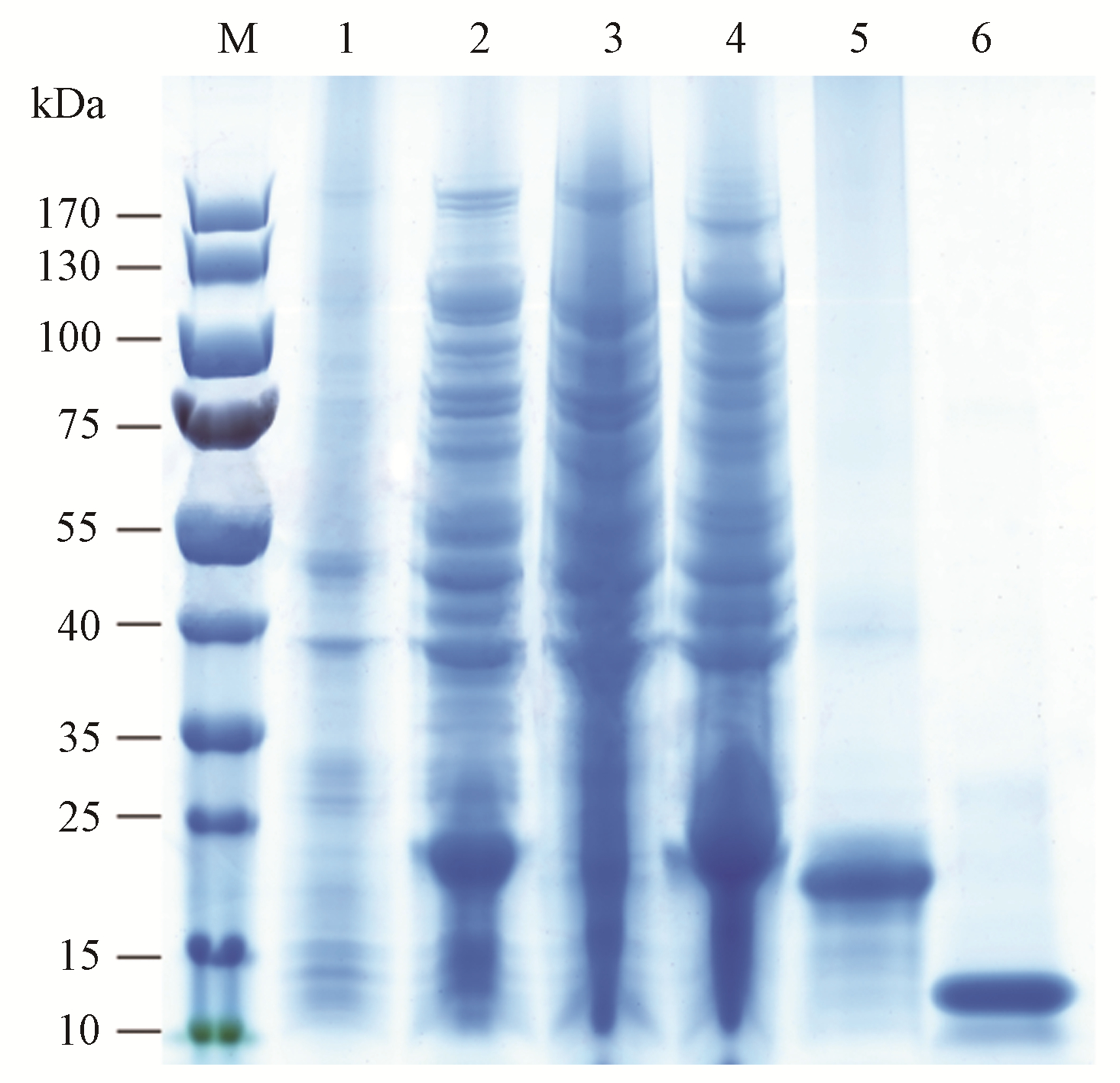
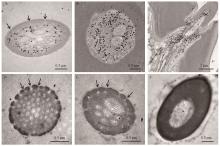
图3
AplaOBP2蛋白在白蜡窄吉丁触角的免疫定位 A: 黑色颗粒标记的AplaOBP2蛋白分布在锥形感器类型I的感器腔中Black spots labeled AplaOBP2 proteins were distributed in the hair lumen of the sensilla basiconica type I;横切(B)和纵切(C)显示AplaOBP2蛋白在锥形感器I基部表达A cross section(B) and longitudinal section(C) showed the expression of AplaOBP2 proteins at the base of s. basiconica type I;锥形感器Ⅱ(D)、锥形感器Ⅲ(E)和单孔化学感器(F) 的感器腔未被标记The hair lumen of s. basiconica type Ⅱ(D), s. basiconica type Ⅲ (E) and uniporous chemoreceptors (F) was never labeled;箭头表示嗅觉感器上的壁孔Arrows indicate wall pores on the sensillum."

表2
白蜡窄吉丁雌雄成虫对AplaOBP2配体(10 mg·mL-1) 的EAG反应①"
| 化合物 Chemical | 标准化EAG Normalized EAG | |
| 雌虫Female | 雄虫Male | |
| 反-2-己烯醛 trans-2-hexenal | 0.441±0.045 abA | 0.533±0.116 aA |
| 反-2-庚烯醛 trans-2-heptenal | 0.613±0.097 aA | 0.509±0.074 aA |
| 苯甲醛 Benzaldehyde | 0.270±0.064 bcA | 0.192±0.074 bA |
| 4′-乙基苯乙酮 4′-ethylacetophenone | 0.182±0.093 bcA | 0.130±0.046 bA |
| 3′, 4′-二甲基苯乙酮 3′, 4′-dimethylacetophenone | 0.091±0.037 cA | 0.146±0.032 bA |
| β-紫罗兰酮 β-ionone | 0.027±0.031 cA | 0.049±0.025 bA |
| 杨叶青, 王山宁, 彭勇, 等. 气味结合蛋白MmedOBP19在中红侧沟茧蜂足部的表达及配体结合特征. 昆虫学报, 2017, 60 (6): 613- 620. | |
| Yang Y Q , Wang S N , Peng Y , et al. Expression of the odor binding protein MmedOBP19 in the legs of Microplitis mediator(Hymenoptera: Braconidae) and its ligand binding characteristics. Acta Entomologica Sinica, 2017, 60 (6): 613- 620. | |
|
Andersson M N , Keeling C I , Mitchell R F . Genomic content of chemosensory genes correlates with host range in wood-boring beetles(Dendroctonus ponderosae, Agrilus planipennis, and Anoplophora glabripennis). BMC Genomics, 2019, 20 (1): 1- 17.
doi: 10.1186/s12864-018-5379-1 |
|
|
Binyameen M , Anderson P , Ignell R , et al. Spatial organization of antennal olfactory sensory neurons in the female Spodoptera littoralis moth: differences in sensitivity and temporal characteristics. Chemical Senses, 2012, 37 (7): 613- 629.
doi: 10.1093/chemse/bjs043 |
|
| Bradford M M . A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 1976, 72 (1/2): 248- 254. | |
|
Bruce T J , Wadhams L J , Woodcock C M . Insect host location: a volatile situation. Trends in Plant Science, 2005, 10 (6): 269- 274.
doi: 10.1016/j.tplants.2005.04.003 |
|
|
Cáceres L A , Lakshminarayan S , Yeung K C , et al. Repellent and attractive effects of α-, β-, and Dihydro-β-lonone to generalist and specialist herbivores. Journal of Chemical Ecology, 2016, 42 (2): 107- 117.
doi: 10.1007/s10886-016-0669-z |
|
|
Crook D J , Khrimian A , Francese J A , et al. Development of a host-based semiochemical lure for trapping emerald ash borer Agrilus planipennis(Coleoptera: Buprestidae). Environmental Entomology, 2008a, 37 (2): 356- 365.
doi: 10.1093/ee/37.2.356 |
|
|
Crook D J , Kerr L M , Mastro V C . Distribution and fine structure of antennal sensilla in emerald ash borer(Coleoptera: Buprestidae). Annals of the Entomological Society America, 2008b, 101 (6): 1103- 1111.
doi: 10.1603/0013-8746-101.6.1103 |
|
|
de Groot P , Grant G G , Poland T M , et al. Electrophysiological response and attraction of emerald ash borer to green leaf volatiles(GLVs) emitted by host foliage. Journal of Chemical Ecology, 2008, 34 (9): 1170.
doi: 10.1007/s10886-008-9514-3 |
|
|
Dippel S , Oberhofer G , Kahnt J , et al. Tissue-specific transcriptomics, chromosomal localization, and phylogeny of chemosensory and odorant binding proteins from the red flour beetle Tribolium castaneum reveal subgroup specificities for olfaction or more general functions. BMC Genomics, 2014, 15, 1141.
doi: 10.1186/1471-2164-15-1141 |
|
|
Forêt S , Maleszka R . Function and evolution of a gene family encoding odorant binding-like proteins in a social insect, the honey bee(Apis mellifera). Genome Research, 2006, 16 (11): 1404- 1413.
doi: 10.1101/gr.5075706 |
|
|
Grant G G , Poland T M , Tina C , et al. Comparison of male and female emerald ash borer(Coleoptera: Buprestidae) responses to phoebe oil and(Z)-3-hexenol lures in light green prism traps. Journal of Economic Entomology, 2011, 104 (1): 173- 179.
doi: 10.1603/EC10197 |
|
| Grant G G , Ryall K L , Lyons D B , et al. Differential response of male and female emerald ash borers(Col., Buprestidae) to(Z)-3-hexenol and manuka oil. Journal of Applied Entomolology, 2010, 134 (1): 26- 33. | |
|
Gu S H , Wang W X , Wang G R , et al. Functional characterization and immunolocalization of odorant binding protein 1 in the lucerne plant bug, Adelphocoris lineolatus (GOEZE). Archives of Insect Biochemistry and Physiology, 2011, 77 (2): 81- 99.
doi: 10.1002/arch.20427 |
|
|
Hansson B S , Stensmyr M C . Evolution of insect olfaction. Neuron, 2011, 72 (5): 698- 711.
doi: 10.1016/j.neuron.2011.11.003 |
|
|
Hekmat-Scafe D S , Scafe C R , McKinney A J , et al. Genome-wide analysis of the odorant-binding protein gene family in Drosophila melanogaster. Genome Research, 2002, 12 (9): 1357- 1369.
doi: 10.1101/gr.239402 |
|
|
Herms D A , McCullough D G . Emerald ash borer invasion of North America: history, biology, ecology, impacts, and management. Annual Review Entomology, 2014, 59 (1): 13- 30.
doi: 10.1146/annurev-ento-011613-162051 |
|
|
Huang G Z , Liu J T , Zhou J J , et al. Expressional and functional comparisons of two general odorant binding proteins in Agrotis ipsilon. Insect Biochemistry and Molecular Biology, 2018, 98, 34- 47.
doi: 10.1016/j.ibmb.2018.05.003 |
|
|
Jiang X C , Ryl M , Krieger J , et al. Odorant binding proteins of the desert locust Schistocerca gregaria(Orthoptera, Acrididae): topographic expression patterns in the antennae. Frontiers in Physiology, 2018, 9, 417.
doi: 10.3389/fphys.2018.00417 |
|
|
Krieger J , Breer H . Olfactory reception in invertebrates. Science, 1999, 286 (5440): 720- 723.
doi: 10.1126/science.286.5440.720 |
|
|
Larter N K , Sun J S , Carlson J R . Organization and function of Drosophila odorant binding proteins. eLife, 2016, 5, e20242.
doi: 10.7554/eLife.20242 |
|
| Laue M , Steinbrecht R A , Ziegelberger G . Immunocytochemical localization of general odorant-binding protein in olfactory sensilla of the silkmoth Antheraea polyphemus. Naturwissenschaften, 1994, 81 (4): 178- 180. | |
|
Leal W S . Odorant reception in insects: roles of receptors, binding proteins, and degrading enzymes. Annual Review of Entomology, 2013, 58 (1): 373- 391.
doi: 10.1146/annurev-ento-120811-153635 |
|
|
Mamidala P , Wijeratne A J , Wijeratne S , et al. Identification of odor-processing genes in the emerald ash borer, Agrilus planipennis. PLoS ONE, 2013, 8 (2): e56555.
doi: 10.1371/journal.pone.0056555 |
|
|
McKenzie S K , Oxley P R , Kronauer D J . Comparative genomics and transcriptomics in ants provide new insights into the evolution and function of odorant binding and chemosensory proteins. BMC Genomics, 2014, 15 (1): 718.
doi: 10.1186/1471-2164-15-718 |
|
|
Morin R S , Liebhold A M , Pugh S A , et al. Regional assessment of emerald ash borer, Agrilus planipennis, impacts in forests of the Eastern United States. Biological Invasions, 2017, 19 (2): 703- 711.
doi: 10.1007/s10530-016-1296-x |
|
|
Pelletier J , Guidolin A , Syed Z , et al. Knockdown of a mosquito odorant-binding protein involved in the sensitive detection of oviposition attractants. Journal of Chemical Ecology, 2010, 36 (3): 245- 248.
doi: 10.1007/s10886-010-9762-x |
|
| Pelosi P , Iovinella I , Felicioli A , et al. Soluble proteins of chemical communication: an overview across arthropods. Frontiers in Physiology, 2014, 5, 320. | |
| Pelosi P , Iovinella I , Zhu J , et al. Beyond chemoreception: Diverse tasks of soluble olfactory proteins in insects. Biological Reviews of the Cambridge Philosophical Society, 2017, 93 (1): 184- 200. | |
|
Pelosi P . Perireceptor events in olfaction. J Neurobiol, 1996, 30 (1): 3- 19.
doi: 10.1002/(SICI)1097-4695(199605)30:1<3::AID-NEU2>3.0.CO;2-A |
|
|
Pikielny C W , Hasan G , Rouyer F , et al. Members of a family of Drosophila putative odorant-binding proteins are expressed in different subsets of olfactory hairs. Neuron, 1994, 12 (1): 35- 49.
doi: 10.1016/0896-6273(94)90150-3 |
|
|
Rigsby C M , Mccartney N B , Herms D A , et al. Variation in the volatile profiles of black and manchurian ash in relation to emerald ash borer oviposition preferences. Journal of Chemical Ecology, 2017, 43 (8): 831- 842.
doi: 10.1007/s10886-017-0873-5 |
|
|
Rodriguez-Saona C , Poland T M , Miller J R , et al. Behavioral and electrophysiological responses of the emerald ash borer, Agrilus planipennis, to induced volatiles of manchurian ash, Fraxinus mandshurica. Chemoecology, 2006, 16 (2): 75- 86.
doi: 10.1007/s00049-005-0329-1 |
|
|
Silk P J , Ryall K , Barry Lyons D , et al. A contact sex pheromone component of the emerald ash borer Agrilus planipennis Fairmaire(Coleoptera: Buprestidae). Naturwissenschaften, 2009, 96 (5): 601- 608.
doi: 10.1007/s00114-009-0513-1 |
|
|
Steinbrecht R A , Laue M , Ziegelberger G . Immunolocalization of pheromone-binding protein and general odorant-binding protein in olfactory sensilla of the silk moths Antheraea and Bombyx. Cell and Tissue Research, 1995, 282 (2): 203- 217.
doi: 10.1007/BF00319112 |
|
| Steinbrecht R A . Pore structures in insect olfactory sensilla: a review of data and concepts. International Journal of Insect Morphology Embryology, 1997, 26 (3/4): 229- 245. | |
|
Sun X , Zhao Z F , Zeng F F , et al. Functional characterization of a pheromone-binding protein from rice leaffolder Cnaphalocrocis medinalis in detecting pheromones and host plant volatiles. Bulletin Entomological Research, 2016, 106 (6): 781- 789.
doi: 10.1017/S0007485316000560 |
|
| Takken W . The role of olfaction in host-seeking of mosquitoes: a review. International Journal of Tropical Insect Science, 1991, 12 (1-3): 287- 295. | |
|
Vet L E M , Dicke M . Ecology of infochemical use by natural enemies in a tritrophic context. Annual Review of Entomology, 1992, 37, 141- 172.
doi: 10.1146/annurev.en.37.010192.001041 |
|
|
Vieira F G , Rozas J . Comparative genomics of the odorant-binding and chemosensory protein gene families across the Arthropoda: origin and evolutionary history of the chemosensory system. Genome Biology Evolouion, 2011, 3, 476- 490.
doi: 10.1093/gbe/evr033 |
|
|
Visser J H . Host odor perception in phytophagous insects. Annual Review of Entomology, 1986, 31, 121- 144.
doi: 10.1146/annurev.en.31.010186.001005 |
|
|
Vogt R G , Riddiford L M . Pheromone binding and inactivation by moth antennae. Nature, 1981, 293 (5828): 161- 163.
doi: 10.1038/293161a0 |
|
|
Wang S N , Shan S , Liu J T , et al. Characterization of antennal chemosensilla and associated odorant binding as well as chemosensory proteins in the parasitoid wasp Microplitis mediator (Hymenoptera: Braconidae). Scientific Reports, 2018, 8, 7649.
doi: 10.1038/s41598-018-25996-3 |
|
|
Wang S N , Shan S , Yu G Y , et al. Identification of odorant-binding proteins and functional analysis of antenna-specific AplaOBP1 in the emerald ash borer, Agrilus planipennis. Journal of Pest Science, 2020, 93 (2): 853- 865.
doi: 10.1007/s10340-019-01188-4 |
|
|
Wilson R I , Mainen Z F . Early events in olfactory processing. Annual Review of Neuroscience, 2006, 29, 163- 201.
doi: 10.1146/annurev.neuro.29.051605.112950 |
|
|
Xu P X , Atkinson R , Jones D N , et al. Drosophila OBP LUSH is required for activity of pheromone-sensitive neurons. Neuron, 2005, 45 (2): 193- 200.
doi: 10.1016/j.neuron.2004.12.031 |
|
|
Ye Z F , Liu X L , Han Q , et al. Functional characterization of PBP1 gene in Helicoverpa armigera(Lepidoptera: Noctuidae) by using the CRISPR/Cas9 system. Scientific Reports, 2017, 7 (1): 8470.
doi: 10.1038/s41598-017-08769-2 |
|
|
Zacharuk R Y . Ultrastructure and function of insect chemosensilla. Annual Review Entomology, 1980, 25, 27- 47.
doi: 10.1146/annurev.en.25.010180.000331 |
| [1] | 党英侨,张彦龙,王小艺,魏可,杨忠岐. 白蜡窄吉丁对高温高湿的耐受性[J]. 林业科学, 2021, 57(3): 189-194. |
| [2] | 宋丽文;李兴鹏;张健;陈越渠;左彤彤. 针叶树小蠹对非寄主挥发物的嗅觉识别和行为反应研究评述[J]. 林业科学, 2012, 48(12): 93-100. |
| [3] | 程彬;付晓霞;韩启;张宝民;张大明;李兴鹏;高长启;孙晓玲. 虫害诱导的家榆挥发物对榆紫叶甲寄主选择行为的影响[J]. 林业科学, 2010, 46(10): 76-82. |
| [4] | 张振;迟德富;宇佳;李晓灿;赵晓杰. 青杨脊虎天牛对13种植物挥发物的电生理及行为反应[J]. 林业科学, 2010, 46(10): 69-75. |
| [5] | 严善春杨慧 高璐璐 王志波 毛洪波. 兴安落叶松鞘蛾对寄主挥发物的反应[J]. 林业科学, 2009, 12(5): 94-101. |
| [6] | 程立超 迟德富 谢兴 王广利. 杨树树皮挥发物的超临界萃取及对青杨脊虎天牛行为的影响*[J]. 林业科学, 2009, 12(11): 109-114. |
| [7] | 李 娟 王满张志春 陈京元 张国安. 云斑天牛成虫对植物气味的行为反应*[J]. 林业科学, 2008, 44(6): 171-175. |
| [8] | 张风娟;武晓颖 杨莉 金幼菊. 超临界CO2萃取五角枫挥发物及其对光肩星天牛的嗅觉行为反应[J]. 林业科学, 2007, 43(6): 146-150. |
| [9] | 王小艺 杨忠岐 刘桂军 刘恩山. 白蜡窄吉丁幼虫的龄数和龄期测定[J]. 林业科学, 2005, 41(3): 97-102. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||