

林业科学 ›› 2021, Vol. 57 ›› Issue (6): 74-84.doi: 10.11707/j.1001-7488.20210608
杨晓红1,陈晓阳2
收稿日期:2020-08-20
出版日期:2021-06-25
发布日期:2021-08-06
基金资助:Xiaohong Yang1,Xiaoyang Chen2
Received:2020-08-20
Online:2021-06-25
Published:2021-08-06
摘要:
目的: 建立安全、无抗生素的二色胡枝子遗传转化体系,通过基因工程技术进一步提高二色胡枝子的抗逆性。方法: 采用PCR法从大肠杆菌DH5α菌株中克隆木糖异构酶基因(xylA);构建含有xylA和甜菜碱醛脱氢酶基因(BADH)的植物表达载体pBI121-xylA-BADH;培养基中用木糖取代一定量的蔗糖建立二色胡枝子的木糖选择系统;以农杆菌介导法对二色胡枝子的子叶节进行遗传转化,通过PCR和Southern检测确定转基因植株;对转化植株及非转化植株进行0.5~2 g·L-1NaCl胁迫,观测植株生长表现,测定1 g·L-1NaCl胁迫下转化植株和非转化植株的甜菜碱醛脱氢酶活性、叶绿素、电导率。提取转化植株及非转化植株组培苗叶片中的可溶性总糖,用高效液相色谱仪分析糖成分。结果: 测序结果表明,克隆的木糖异构酶基因与GenBank公布的大肠杆菌xylA核苷酸序列的同源性达到100%,PCR及酶切检测表明正确构建了pBI121-xylA-BADH植物表达载体。在二色胡枝子遗传转化过程中,应在纯木糖培养基上筛选抗性芽,在抗性芽增殖和生根过程中,可以采用蔗糖5 g·L-1、木糖25 g·L-1的糖类混合剂进行进一步筛选。在农杆菌介导的遗传转化中,外植体预培养1天后,采用LBA4404菌株侵染20~30 min,共培养3天,可以获得相对较多的木糖抗性芽。对获得的部分抗性芽进行PCR及Southern检测,得到2个转基因株系,二色胡枝子遗传转化率低于4.7%。耐盐性观测表明,转基因植株能够在含有2 g·L-1NaCl的培养基上生长并生根,而非转基因植株在相同培养基上则叶子变黄、脱落,且不能生根。无盐胁迫时,转基因和非转基因植株在甜菜碱醛脱氢酶活性、叶绿素含量和电导率方面无显著性差异;盐胁迫后,非转基因植株的甜菜碱醛脱氢酶活性无明显变化,而转基因植株的甜菜碱醛脱氢酶活性大幅度提升,且达到非转基因植株的8~10倍;盐胁迫后,转基因植株的叶绿素含量显著高于非转基因植株,而电导率则显著低于非转基因植株。高效液相色谱的分析结果显示,转基因植株叶片中测出了果糖和葡萄糖,而非转基因植株叶片中测出了葡萄糖和一种未标示的糖类。结论: 建立了二色胡枝子在木糖选择系统下的遗传转化体系,并获得稳定表达的转基因植株。外源甜菜碱醛脱氢酶基因可提高二色胡枝子的耐盐性;来源于大肠杆菌的木糖异构酶基因的导入会影响二色胡枝子的糖代谢。
中图分类号:
杨晓红,陈晓阳. 木糖选择系统下xylA & BADH双价基因对二色胡枝子的遗传转化[J]. 林业科学, 2021, 57(6): 74-84.
Xiaohong Yang,Xiaoyang Chen. Transformation of Lespedeza bicolor with Bivalent Gene xylA & BADH under Xylose Selection System[J]. Scientia Silvae Sinicae, 2021, 57(6): 74-84.
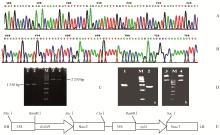
图1
xylA克隆和pBI121-xylA-BADH植物表达载体的构建 A: 克隆出的xylA基因测序的部分片段;B: 35S-BADH-nos测序的部分片段;C: pBI121-xylA载体的PCR检测(M: DNA marker;1, 2:不同连接产物的xylA检测;3, 4: 不同连接产物的35S-xylA-nos片段检测);D: pBI121-xylA-BADH的PCR检测及酶切检测[a: PCR检测;b: BamHⅠ酶切检测;1: 检测BADH片段(1 520 bp);2: 检测xylA片段(1 350 bp);3, 4: BamHⅠ酶切结果;M: DNA marker];E: pBI121-xylA-BADH植物表达载体。"

表1
木糖对二色胡枝子子叶节再生、茎段分化及组培苗生根的影响①"
| 糖种类及含量 Kind and contents of saccharides | 子叶节Cotyledonary node | 茎段Shoot segment | 组培苗生根Rooting in vitro | |||||
| 分化率 Differentiation rate (%) | 平均分化不定芽数 Average bud number | 分化率 Differentiation rate (%) | 平均分化不定芽数 Average bud number | 生根率 Rooting percentage (%) | 平均生根数 Average root number | |||
| CK(蔗糖30 g·L-1) (Sucrose 30 g·L-1) | 100.0±3.4 a | 4.2±0.16 a | 94.0±2.63 a | 2.79±0.20 a | 92.0±9.93 a | 2.90±0.11 a | ||
| 木糖15 g·L-1+蔗糖15 g·L-1 Xylose 15 g·L-1+sucrose 15 g·L-1 | 90.0±5.9 b | 0.9±0.12 b | 22.0±1.28 b | 0.48±0.01 b | 42.0±3.57 b | 0.86±0.14 b | ||
| 木糖20 g·L-1+蔗糖10 g·L-1 Xylose 20 g·L-1+sucrose 10 g·L-1 | 68.0±4.6 c | 0.3±0.11 c | 12.0±0.01 c | 0.10±0.03 c | 40.0±0.24 b | 0.64±0.28 b | ||
| 木糖25 g·L-1+蔗糖5 g·L-1 Xylose 25 g·L-1+sucrose 5 g·L-1 | 52.0±2.4 d | 0.1±0.01 c | 0.0±0.00 d | 0.00±0.00 c | 0.0±0.00 c | 0.00±0.00 c | ||
| 木糖30 g·L-1 Xylose 30 g·L-1 | 2.0±0.1 e | 0.0±0.00 c | 0.0±0.00 d | 0.00±0.00 c | 0.0±0.00 c | 0.00±0.00 c | ||
表4
NaCl胁迫对转xylA & BADH基因二色胡枝子生理指标的影响"
| 处理 Treatment | 株系 Line | BADH活性 BADH activity/(mmol·mg-1min-1) | 叶绿素 Chlorophyll/(mg·g-1FW) | 电导率 Electric conductivity (%) |
| 无盐处理 Without NaCl stress | CK | 0.072 6±0.036 a | 4.71±0.16 a | 11.041±0.088 a |
| 株1 Line 1 | 0.144 8±0.019 a | 4.85±0.20 a | 11.570±0.137 a | |
| 株2 Line 2 | 0.113 5±0.025 a | 5.07±0.25 a | 11.521±0.076 a | |
| NaCl处理 Under NaCl stress | CK | 0.128±0.029 a | 3.01±0.03 a | 58.106±0.049 a |
| 株1 Line 1 | 1.369±0.133 b | 4.25±0.08 b | 32.232±0.043 b | |
| 株2 Line 2 | 1.067±0.213 b | 4.37±0.04 b | 35.519±0.042 b |
| 丁霄, 隋炯明, 王晶珊, 等. 以木糖异构酶基因作为选择标记的花生遗传转化. 核农学报, 2012, 26 (3): 444- 448. | |
| Ding X , Sui J M , Wang J S , et al. The genetic transformation of peanut using xylose isomerase gene as a selection marker. Journal of Nuclear Agricultural Sciences, 2012, 26 (3): 444- 448. | |
| 郭新梅, 张晓东, 梁荣奇, 等. 以木糖异构酶基因为筛选标记的玉米遗传转化. 植物生理与分子生物学报, 2007, 33 (6): 547- 552. | |
| Gou X M , Zhang X D , Liang R Q , et al. Maize transformation using xylose isomerase gene as a selection marker. Journal of Plant Physiology and Molecular Biology, 2007, 33 (6): 547- 552. | |
| 黄天伟. 2009. 果聚糖蔗糖转移酶(SacB)基因转化二色胡枝子的研究. 北京: 北京林业大学硕士学位论文, 19-28. | |
| Huang T W. 2009. Studies on transformation of Lespedeza bicolor with SacB gene. Beijing: MS thesis of Beijing Forestry University, 19-28. [in Chinese] | |
| 穆永光. 2007. 非盐碱生境的二种豆科灌木盐碱耐性研究. 长春: 东北师范大学硕士学位论文, 11-14. | |
| Mu Y G. 2007. Study on saline-alkali tolerance of two species of leguminous shrubs in non-saline-alkali habitats. Changchun: MS thesis of Northeast Normal University, 11-14. [in Chinese] | |
| 王婷, 李进军, 吴跃明, 等. 2008. 甘露糖阳性选择系统的建立及在紫花苜蓿转基因中的应用//中国畜牧兽医学会动物营养学分会第十次学术研讨会论文集. 北京: 中国农业科学技术出版社. | |
| Wang T, Li J J, Wu Y M, et al. 2008. Establishment of mannose positive selection system and its application in transformation of Medicago sativa L. //Proceedings of the 10th symposium of Animal Nutrition Branch, Chinese Society of Animal Husbandry and Veterinary Science. Beijing: China Agricultural Science and Technology Press. [in Chinese] | |
| 韦正乙. 2007. 利用木糖选择系统获得HAL1转基因百脉根(Lotus cirniculatus)植株及耐盐性鉴定. 长春: 东北师范大学硕士学位论文, 1-4. | |
| Wei Z Y. 2007. Getting HAL1 transgenic plants of Lotus cirniculatus by using xylose selective system and identification for salt tolerance. Changchun: MS thesis of Northeast Normal University, 1-4. [in Chinese] | |
| 解小娟, 杨晓红, 陈晓阳. 遮荫对转BADH基因的美丽胡枝子叶片形态和光合特性的影响. 林业科学, 2013, 49 (3): 33- 42. | |
| Xie X J , Yang X H , Chen X Y . Effects of shading on leaf shape and photosynthetic characteristics of the transgenic Lespedeza formosa with expressing BADH gene. Scientia Silvae Sinicae, 2013, 49 (3): 33- 42. | |
|
谢鑫星, 路扬, 梁晶, 等. 高效农杆菌介导的紫花苜蓿遗传转化体系的建立. 中国农业科技导报, 2010, 12 (1): 128- 134.
doi: 10.3969/j.issn.1008-0864.2010.01.22 |
|
|
Xie X X , Lu Y , Liang J , et al. Establishment of an efficient Agrobacterium-mediated transformation system for genetic improvement of Alfalfa (Medicago sativaL). Journal of Agricultural Science and Technology, 2010, 12 (1): 128- 134.
doi: 10.3969/j.issn.1008-0864.2010.01.22 |
|
| 闫新甫, 李润植, 苗泽伟, 等. 转基因植物. 北京: 科学出版社, 2003: 154- 193. | |
| Yan X P , Li R Z , Miao Z W , et al. Transgenic plant. Beijing: Science Press, 2003: 154- 193. | |
| 杨晓红, 付宇辰, 张民民, 等. 2个胡枝子新品种嫩枝扦插繁殖技术研究. 江西农业大学学报, 2019, 41 (1): 99- 106. | |
| Yang X H , Fu Y C , Zhang M M , et al. A study of the techniques for the softwood cutting propagation of two new varieties of Lespedeza bicolor Turcz. Acta Agriculturae Universitatis Jiangxiensis, 2019, 41 (1): 99- 106. | |
|
叶纨芝, 曹家树, 曾广文. 基因工程植物商品化生产面临的问题与前景. 植物学通报, 2000, 17 (5): 394- 400.
doi: 10.3969/j.issn.1674-3466.2000.05.002 |
|
|
Ye W Z , Cao J S , Zeng G W . Issues and challenges about plant gene engineering to commercial production. Chinese Bulletin of Botany, 2000, 17 (5): 394- 400.
doi: 10.3969/j.issn.1674-3466.2000.05.002 |
|
|
张宁, 司怀军, 李学才, 等. 根癌农杆菌介导的马铃薯高效遗传转化体系的研究. 中国马铃薯, 2004, 18 (3): 132- 135.
doi: 10.3969/j.issn.1672-3635.2004.03.002 |
|
| Zhang N, Si H J, Li X C, et al. An efficient transformation system of potato mediated by Agrobacterium tumefaciens. Chinese Potato Journal, 18(3): 132-135. [in Chinese] | |
|
赵建华, 李浩霞, 尹跃, 等. 枸杞木糖异构酶基因LbxylA的克隆、原核表达及多克隆抗体的制备. 食品科学, 2019, 40 (10): 77- 83.
doi: 10.7506/spkx1002-6630-20180528-382 |
|
|
Zhao J H , Li H X , Yin Y , et al. Cloning and prokaryotic expression of the xylose isomerase gene(LbxylA) from Chinese wolfberry (Lycium barbarum Linn. ) fruit and preparation of polyclonal antibody against the fused protein. Food Science, 2019, 40 (10): 77- 83.
doi: 10.7506/spkx1002-6630-20180528-382 |
|
| 朱军, 韩锁义, 袁美, 等. 农杆菌介导的花生遗传转化条件优化. 中国油料作物学报, 2018, 40 (2): 191- 198. | |
| Zhu J , Han S Y , Yuan M , et al. Optimization of Agrobacterium tumefaciens-mediated transformation in peanut. Chinese Journal of Oil Crop Science, 2018, 40 (2): 191- 198. | |
|
Bříza J , Pavingerová D , Přikrylová P , et al. Use of phosphomannose isomerase-based selection system for Agrobacterium-mediated transformation of tomato and potato. Biologia Plantarum, 2008, 52 (3): 453- 461.
doi: 10.1007/s10535-008-0090-8 |
|
|
Chen T H H , Murata N . Glycinebetaine: an effective protectant against abiotic stress in plants. Trends in Plant Science, 2008, 13 (9): 499- 505.
doi: 10.1016/j.tplants.2008.06.007 |
|
|
Chen T H H , Murata N . Glycinebetaine protects plants against abiotic stress: mechanisms and biotechnological applications. Plant, Cell and Environment, 2011, 34 (1): 1- 20.
doi: 10.1111/j.1365-3040.2010.02232.x |
|
|
Daniell H , Muthukumar B , Lee S B . Marker free transgenic plants: engineering the chloroplast genome without the use of antibiotic selection. Current Genetics, 2001, 39 (2): 109- 116.
doi: 10.1007/s002940100185 |
|
|
Egnin M , Mora A , Prakash C S . Factors enhancing Agrobacterium tumefaciens-mediated gene transfer in peanut (Arachis hypogaea L. ). In Vitro Cellular & Developmental Biology-Plant, 1998, 34 (4): 310- 318.
doi: 10.1007/BF02822740 |
|
| Erlich H A . PCR Technology. New York: Stockon Press, 1989: 17- 22. | |
|
Ewa U W , Andrea L , Uta R T , et al. Expression of a bacterial xylose isomerase in potato tubers results in an altered hexose composition and a consequent induction of metabolism. Plant Cell Physiology, 2003, 44 (12): 1359- 1367.
doi: 10.1093/pcp/pcg166 |
|
|
Haldrup A , Petersen S G , Okkels F T . Positive selection: a plant selection principle based on xylose isomerase, an enzyme used in the food industry. Plant Cell Reports, 1998a, 18, 76- 81.
doi: 10.1007/s002990050535 |
|
|
Haldrup A , Petersen S G , Okkels F T . The xylose isomerase gene from Thermoanaerobacterium thermosulfurogenes allows effective selection of transgenic plant cells using D-xylose as the selection agent. Plant Molecular Biology, 1998b, 37, 287- 296.
doi: 10.1023/A:1005910417789 |
|
|
Haldrup A , Noerremark M , Okkels F T . Plant selection principle based on xylose isomerase. In Vitro Cell Development, 2001, 37 (2): 114- 119.
doi: 10.1007/s11627-001-0022-1 |
|
| Ishida Y , Saito H , Ohta S , et al. High efficiency transformation of maize(Zea mays L. ) mediated by Agrobacterium tumefaciens. Nature Biotechnology, 1996, 14, 745- 750. | |
|
Kristo P , Saarelainen R , Fagerström R , et al. Protein purification, and cloning and characterization of the cDNA and gene for xylose isomerase of barley. European Journal of Biochemistry, 1996, 237 (1): 240- 246.
doi: 10.1111/j.1432-1033.1996.0240n.x |
|
|
Lilius G , Mejare A , Hofvander P , et al. Enhanced fructose levels in field grown potato tubers expressing a thermostable glucose isomerase. Biotechnology Letters, 2000, 22, 1035- 1041.
doi: 10.1023/A:1005618110912 |
|
| McNeil S D , Nuccio M L , Hanson A D . Betaines and related osmoprotectants. Targets for metabolic engineering of stress resistance. Journal Plant Physiology, 1999, 120 (4): 945- 950. | |
| Mert M J , Rose S H , La Grange D C , et al. Quantitative metabolomics of a xylose-utilizing Saccharomyces cerevisiae strain expressing the Bacteroides thetaiotaomicron xylose isomerase on glucose and xylose. Journal of Industrial Microbiology & Biotechnology, 2017, 44 (10): 1459- 1470. | |
|
Miki B , McHugh S . Selectable marker genes in transgenic plants: applications, alternatives and biosafety. Journal of Biotechnology, 2004, 107 (3): 193- 232.
doi: 10.1016/j.jbiotec.2003.10.011 |
|
|
Negrotto D , Jolley M , Beer S , et al. The use of phosphomannose-isomerase as a selectable marker to recover transgenic maize plants (Zea mays L. ) via Agrobacterium transformation. Plant Cell Reports, 2000, 19, 798- 803.
doi: 10.1007/s002999900187 |
|
| Rhodes D , Hanson A D . Quatemary ammonium and tertiary sulfonium compounds in higher plants. Annual Review of Plant Physiology and Plant Molecular Biology, 2003, 44 (1): 357- 384. | |
|
Sakamoto A , Murata N . Genetic engineering of glycinebetaine synthesis in plants: current status and implications for enhancement of stress tolerance. Journal of Experimental Botany, 2000, 51, 81- 88.
doi: 10.1093/jexbot/51.342.81 |
|
|
Sathya R , Mohan R S , Varalakshmi P , et al. Purification and kinetic behavior of glucose isomerase from Streptomyces lividans RSU26. Saudi Journal of Biological Sciences, 2020, 27 (4): 1117- 1123.
doi: 10.1016/j.sjbs.2019.12.024 |
|
| Takabe T, Rai V, Hibino T. 2006. Metabolic engineering of glycinebetaine//Takabe T, Rai V, Hibino T. Abiotic stress tolerance in plants. Dordrecht, The Netherlands: Springer, 137-151. | |
| Van Maris A J , Winkler A A , Kuyper M , et al. Development of efficient xylose fermentation in Saccharomyces cerevisiae: xylose isomerase as a key component. Advances in Biochemical Engineering/Biotechnology, 2008, 108, 179- 204. |
| [1] | 刘闵豪,徐郡儡,叶靖,李周岐,范睿深,李龙. 农杆菌介导的杜仲叶片愈伤组织遗传转化体系[J]. 林业科学, 2020, 56(2): 79-88. |
| [2] | 梁璧,张佳琦,任飞,胡恒康,徐川梅,胡渊渊,黄有军,娄和强,张启香. 山核桃贝壳杉烯氧化酶基因CcKO的克隆和表达分析[J]. 林业科学, 2020, 56(10): 70-82. |
| [3] | 谷战英, 杨若楠, 陈昊. 油桐叶肉细胞原生质体分离及瞬时转化体系的建立[J]. 林业科学, 2018, 54(1): 46-53. |
| [4] | 程珍霞, 胡海涛, 杨莉, 王长春, 郭卫东, 杨玲. 超表达牛奶子EutPDS提高番茄果实番茄红素含量[J]. 林业科学, 2017, 53(1): 62-69. |
| [5] | 罗在柒, 郭辉力, 杨亚东, 杨明峰, 马兰青, 王有年. 农杆菌介导虎杖芪合酶基因遗传转化壶瓶枣的研究[J]. 林业科学, 2015, 51(10): 101-109. |
| [6] | 张伟溪, 刘浡洋, 丁昌俊, 张冰玉, 黄秦军, 褚延广, 苏晓华. 欧美杨锌指蛋白转录因子基因(ZxZF)的遗传转化及抗旱性初步分析[J]. 林业科学, 2014, 50(3): 31-37. |
| [7] | 李思蒙;王永林;黄冬辉;田呈明. 杨树炭疽病菌原生质体遗传转化的建立及绿色荧光蛋白的表达[J]. , 2013, 49(5): 121-127. |
| [8] | 欧阳乐军;刘媛;黄真池;曾富华. 转TSRF 1基因尾叶桉的培育及其广谱抗病性[J]. 林业科学, 2013, 49(4): 46-53. |
| [9] | 黄艳;刘虹;袁丽;贾之春;罗克明. 过量表达LAR3和Bbchit1以提高毛白杨对叶枯病的抗性[J]. 林业科学, 2012, 48(7): 92-97. |
| [10] | 李晓瑞;胡尚连;曹颖;卢学琴;任鹏;吴晓宇;周美娟. 农杆菌介导慈竹4CL基因遗传转化梁山慈竹[J]. 林业科学, 2012, 48(3): 38-44. |
| [11] | 贾之春;孙一铭;秦志超;肖训焰;齐代华;罗克明. 转抗菌肽基因毛白杨的培育及其对溃疡病的抗性[J]. 林业科学, 2011, 47(7): 123-127. |
| [12] | 贾小明;张焕玲;樊军锋. 热激启动子控制的FT基因诱导杨树早期开花体系的优化[J]. 林业科学, 2011, 47(11): 37-43. |
| [13] | 宋恩慧 蔡诚 魏国 高慧 项艳. RNA干涉培育低木质素杨树[J]. 林业科学, 2010, 46(2): 39-44. |
| [14] | 王曦茁;朴春根;李虹;汪来发;郭民伟;李永;刘晓莉. 根癌农杆菌介导的淡紫拟青霉遗传转化体系的建立[J]. 林业科学, 2010, 46(10): 95-102. |
| [15] | 张晓英.甘敬 尹伟伦 朱祯 王华芳. 国槐遗传转化体系的优化*[J]. 林业科学, 2009, 12(5): 20-26. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||