

林业科学 ›› 2026, Vol. 62 ›› Issue (5): 27-39.doi: 10.11707/j.1001-7488.LYKX20250610
收稿日期:2025-10-10
修回日期:2026-03-30
出版日期:2026-05-10
发布日期:2026-05-12
通讯作者:
韦小丽
E-mail:gdwxl2022@163.com
基金资助:
Yan He1,Nianjie Shang1,2,Shicheng Su1,Xiaoli Wei1,*( )
)
Received:2025-10-10
Revised:2026-03-30
Online:2026-05-10
Published:2026-05-12
Contact:
Xiaoli Wei
E-mail:gdwxl2022@163.com
摘要:
目的: 探究种子际真菌引发对花榈木种子萌发的生理生化机制,为具有硬实特性的林木种子破除休眠提供理论依据和菌种保障。方法: 从花榈木种子际土壤中分离出3株促萌发真菌,测定其促进种皮分解相关酶的分泌能力及内源激素合成量。利用3株真菌引发花榈木种子进行发芽试验,测定其萌发不同阶段的生理生化指标,并以常规方法处理花榈木种子验证引发效果,系统评估3株种子际真菌对花榈木种子萌发的生理调控效应。结果: 1) 菌株SS-1-14、SS-2-3和SS-2-22在培养7天内均能分泌促进种皮分解相关酶。SS-1-14优先降解果胶层,SS-2-3持续释放高活性果胶酶和脂肪酶,SS-2-22则在初期快速产生纤维素酶和果胶酶,高效瓦解种皮屏障。2) 3株菌株均能分泌生长素和细胞分裂素前体,激活种胚细胞分化。SS-1-14产生的吲哚乙酸、水杨酸以及水杨酸葡萄糖苷含量高,兼具促生长与激活系统抗性功能。SS-2-3产吲哚-3-乙腈、3-(2-氨乙基)吲哚和玉米素较多,直接调控细胞分裂和胚芽分化。SS-2-22以生长素类和细胞分裂素前体(N6-异戊烯基腺苷、异戊烯基腺嘌呤)为主,通过前体物质转化合成活性激素,降低代谢负担。3) 种子际真菌引发是通过动态调控花榈木种子的抗氧化酶和贮藏物质代谢,有效促进种子萌发。3株菌株通过调控整个萌发过程中CAT和PPO、后期POD和SOD增强抗氧化能力。SS-1-14菌株引发对种子酶活性的调控效果最佳,可将种子萌发后期SOD以及前、后期CAT活性显著提高19.10%、127.13%和41.20%。3株菌株通过调控整个萌发过程中蛋白酶、脂酶及后期的淀粉酶、酸性磷酸酶活性促进贮藏物质转化,其中SS-1-14作用最为显著,可将种子萌发前、后期的蛋白酶和脂酶分别提高67.31%、58.61%和77.16%、59.32%。3株菌株均可加速整个萌发过程的大分子物质降解以及小分子物质生成,其中SS-1-14引发综合效果最佳,使种子萌发前期甘油三酯、后期淀粉降解率增加32.20%、60.51%,并使种子萌发前期ATP、后期可溶性糖生成分别加快188.83%、65.24%。4) 与刻伤、酸蚀、热水浸泡等传统物理化学处理方法相比,真菌引发可降低种子电导率以及MDA含量,缓解萌发过程中的氧化损伤,维持细胞膜系统的完整性。结论: 种子际真菌通过分泌促进种皮分解酶破除机械障碍、合成植物激素激活种子生理活性,增强抗氧化酶与物质转化酶系统以降低萌发损伤并促进贮藏物质转化,形成“物理?化学?生物”多维度的引发机制,从而促进花榈木硬实种子的萌发。
中图分类号:
何燕,尚念杰,苏石诚,韦小丽. 种子际真菌引发花榈木种子萌发的生理生化机制[J]. 林业科学, 2026, 62(5): 27-39.
Yan He,Nianjie Shang,Shicheng Su,Xiaoli Wei. Physiological and Biochemical Mechanisms of Spermosphere Fungi Induing Germination of Ormosia henryi Seeds[J]. Scientia Silvae Sinicae, 2026, 62(5): 27-39.
表1
种子际真菌对花榈木种子萌发的影响"
| 菌株 Strain | 发芽率 Germination percentage (%) | 发芽势 Germination energy (%) | 发芽指数 Germination index | 平均发芽速率 Mean germination rate/d?1 |
| 对照 Control | 67.29±1.42b | 49.49±0.07bc | 1.88±0.04c | 9.38±1.03a |
| SS-1-14 | 94.44±5.56a | 73.80±0.05a | 3.73±0.10a | 11.77±0.37a |
| SS-2-3 | 83.49±8.94ab | 39.93±0.03c | 3.10±0.26b | 9.68±0.55a |
| SS-2-22 | 90.81±2.93a | 67.27±0.07ab | 3.66±0.07a | 10.92±0.98a |
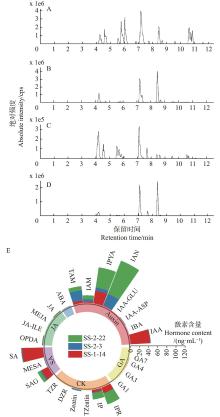
图2
3株种子际真菌的26种植物激素的产生情况 A. 对照组总离子色谱图Total ion chromatograms of control; B. SS-1-14总离子色谱图Total ion chromatograms of SS-1-14; C. SS-2-3总离子色谱图Total ion chromatograms of SS-2-3; D. SS-2-22总离子色谱图Total ion chromatograms of SS-2-22; E. 产生激素种类与含量Types and contents of producing hormones. IAA: 吲哚乙酸Indoleacetic acid; IBA: 吲哚丁酸Indolebutyric acid; IAA-ASP: 吲哚乙酸-天冬氨酸Indoleacetic acid-aspartic acid; IAA-Glu: 吲哚乙酸-谷氨酸Indoleacetic acid-glutamic acid; IAN: 吲哚-3-乙腈Indole-3-acetonitrile; IPYA: 吲哚-3-丙酮酸Indole-3-pyruvic acid; IAM: 吲哚-3-乙酰胺Indole-3-acetamide; TAM: 3-(2-氨乙基) 吲哚Tryptamine; ABA: 脱落酸Abscisic acid; JA: 茉莉酸Jasmonic acid; MEJA: 茉莉酸甲酯Methyl jasmonate; JA-ILE: 茉莉酸-异亮氨酸Jasmonic acid-isoleucine; OPDA: 12-氧代-植物二烯酸12-oxo phytodienoic acid; SA: 水杨酸Salicylic acid; MESA: 水杨酸甲酯Methyl salicylate; SAG: 水杨酸葡萄糖苷Salicylic acid glucoside; TZR: 反玉米素核苷Trans-Zeatin-riboside; DZR: 二氢玉米素核糖Dihidrozeatin riboside; Zeatin玉米素; TZeatin: 反-玉米素Trans-Zeatin; IP: 异戊烯基腺嘌呤Isopentenyladenine; IPR: N6-异戊烯基腺苷N6-isopentenyladenosine; GA1: 赤霉素A1 Gibberellin acid 1; GA3: 赤霉素A3 Gibberellin acid 3; GA4: 赤霉素A4 Gibberellin acid 4; GA7: 赤霉素A7 Gibberellin acid 7."
|
陈美莲, 白睿璇, 林 琳. 产脂肪酶菌株的筛选及鉴定. 福建农业科技, 2023, 54 (9): 63- 68.
doi: 10.16433/j.1673-2383.2021.06.002 |
|
|
Chen M L, Bai R X, Lin L. Screening and identification of lipase-producing strains. Fujian Agricultural Science and Technology, 2023, 54 (9): 63- 68.
doi: 10.16433/j.1673-2383.2021.06.002 |
|
|
陈 瑶, 周寒梅, 何 兵, 等. GA3和IAA组合调控华重楼种子萌发机理初探. 园艺学报, 2020, 47 (2): 321- 333.
doi: 10.16420/j.issn.0513-353x.2019-0263 |
|
|
Chen Y, Zhou H M, He B, et al. Preliminary study on the regulation mechanism of GA3 and IAA combination in Paris polyphylla var. chinensis seed germination. Acta Horticulturae Sinica, 2020, 47 (2): 321- 333.
doi: 10.16420/j.issn.0513-353x.2019-0263 |
|
|
陈艳伟, 韦小丽, 杨玄烨, 等. 珍贵树种木荚红豆硬实种子破除方法研究. 种子, 2015, 34 (11): 37- 40.
doi: 10.16590/j.cnki.1001-4705.2015.11.037 |
|
|
Chen Y W, Wei X L, Yang X Y, et al. Study on breaking methods for hard seed of rare species Ormosia xylocarpa. Seed, 2015, 34 (11): 37- 40.
doi: 10.16590/j.cnki.1001-4705.2015.11.037 |
|
| 陈艳伟. 2016. 红豆树与木荚红豆种子生物学特性及贮藏生理生态. 贵阳: 贵州大学. | |
| Chen Y W. 2016. Biological characteristics and storage eco-physiology of Ormosia hosiei and Ormosia xylocarpa seed. Guiyang: Guizhou University. [in Chinese] | |
| 邓 兆, 韦小丽. 珍稀树种花榈木种子休眠破除方法研究. 种子, 2016, 35 (11): 1- 4. | |
| Deng Z, Wei X L. Study on method of breaking seed dormancy for rare tree species Ormosia henryi. Seed, 2016, 35 (11): 1- 4. | |
|
邓 兆, 韦小丽, 孟宪帅, 等. 花榈木种子休眠和萌发的初步研究. 贵州农业科学, 2011, 39 (5): 69- 72.
doi: 10.3969/j.issn.1001-3601.2011.05.020 |
|
|
Deng Z, Wei X L, Meng X S, et al. A preliminary study on dormancy and germination of Ormosia henryi seeds. Guizhou Agricultural Sciences, 2011, 39 (5): 69- 72.
doi: 10.3969/j.issn.1001-3601.2011.05.020 |
|
| 桂 平, 龙 鹏. 珍稀树种花榈木研究进展. 贵州农业科学, 2021, 49 (7): 98- 106. | |
| Gui P, Long P. Research progress on rare tree species of Ormosia henryi. Guizhou Agricultural Sciences, 2021, 49 (7): 98- 106. | |
| 江瑞敏, 宋曰钦, 胡 重, 等. 2023. 不同处理对花榈木种子萌发影响. 林业科技通讯, (6): 109–112. | |
| Jiang R M, Song Y Q, Hu Z, et al. 2023. Effects of different treatments on seed germination of Ormosia henryi. Forest Science and Technology, (6): 109–112. [in Chinese] | |
| 刘子凡. 2011. 种子学实验指南. 北京: 化学工业出版社. | |
| Liu Z F. 2011. Seed science: a laboratory manual. Beijing: Chemical Industry Press. [in Chinese] | |
| 龙绛雪, 曹基武. 2020. “材貌双全”的花榈木. 中国花卉园艺, (8): 45. | |
| Long J X, Cao J W. 2020. Ormosia henryi, a tree of fine timber and excellent form. China Flowers & Horticulture, (8): 45. [in Chinese] | |
| 卢晓华. 2016. 果胶酶产生菌的筛选鉴定、产酶条件优化及酶学性质研究. 武汉: 湖北工业大学. | |
| Lu X H. 2016. Screen and identification of pectinase-production strains, optimization of pectinase-production conditions and study on characterization of pectinase. Wuhan: Hubei University of Technology. [in Chinese] | |
| 孟雪娇, 邸 昆, 丁国华. 水杨酸在植物体内的生理作用研究进展. 中国农学通报, 2010, 26 (15): 207- 214. | |
| Meng X J, Di K, Ding G H. Progress of study on the physiological role of salicylic acid in plant. Chinese Agricultural Science Bulletin, 2010, 26 (15): 207- 214. | |
|
冉光耀, 唐佳代, 赵益梅, 等. 一株产纤维素酶酵母菌的筛选、鉴定及产酶条件优化. 中国酿造, 2024, 43 (9): 72- 78.
doi: 10.11882/j.issn.0254-5071.2024.09.011 |
|
|
Ran G Y, Tang J D, Zhao Y M, et al. Screening, identification and enzyme production conditions optimization of a cellulase-producing yeast. China Brewing, 2024, 43 (9): 72- 78.
doi: 10.11882/j.issn.0254-5071.2024.09.011 |
|
|
田学军, 卢焕仙. 热胁迫对油菜种子活力和幼苗抗氧化酶活性的影响. 江苏农业科学, 2011, 39 (4): 77- 78.
doi: 10.3969/j.issn.1002-1302.2011.04.028 |
|
|
Tian X J, Lu H X. Effects of heat stress on seed vigor and antioxidant enzyme activity of rape seedlings. Jiangsu Agricultural Sciences, 2011, 39 (4): 77- 78.
doi: 10.3969/j.issn.1002-1302.2011.04.028 |
|
| 王婷婷. 2020. 花榈木种子际促生真菌和内生真菌的分离筛选及其促生效应. 贵阳: 贵州大学. | |
| Wang T T. 2020. Isolation and screening on spermosphere fungi and endophytic fungi in Ormosia henryi seed and their promoting effect for growth. Guiyang: Guizhou University. [in Chinese] | |
|
王小东, 刘 鹏, 刘美娟, 等. 中国红豆属植物生物与生态学特征研究现状. 植物科学学报, 2018, 36 (3): 440- 451.
doi: 10.11913/PSJ.2095-0837.2018.30440 |
|
|
Wang X D, Liu P, Liu M J, et al. Biology and ecology research status of Ormosia species in China. Plant Science Journal, 2018, 36 (3): 440- 451.
doi: 10.11913/PSJ.2095-0837.2018.30440 |
|
| 王学奎. 2006. 植物生理生化实验原理和技术. 北京: 高等教育出版社. | |
| Wang X K. 2006. Principles and techniques of plant physiology and biochemistry experiments. Beijing: Higher Education Press. [in Chinese] | |
| 王 月. 2019. 甲基乙二醛信号诱导玉米幼苗耐热性的形成及其可能的机理. 昆明: 云南师范大学. | |
| Wang Y. 2019. Methylglyoxal signaling induces the heat tolerance of maize seedlings and its possible mechanisms. Kunming: Yunnan Normal University. [in Chinese] | |
| 韦小丽, 孟宪帅, 邓 兆. 珍稀树种花榈木种子繁殖生态学特性与濒危的关系. 种子, 2014, 33 (1): 82- 86. | |
| Wei X L, Meng X S, Deng Z. Relation between being endangered and seed reproductive ecology of a rare species Ormosia henryi. Seed, 2014, 33 (1): 82- 86. | |
| 张 青, 李隆云, 孙年喜. 青蒿种子萌发过程中生理生化变化的研究. 种子, 2011, 30 (3): 10- 13. | |
| Zhang Q, Li L Y, Sun N X. Study on the change of physiology and biochemistry during process of Artemisia annua seed germination. Seed, 2011, 30 (3): 10- 13. | |
| 张志良, 李小方. 2003. 植物生理学实验指导. 北京: 高等教育出版社. | |
| Zhang Z L, Li X F. 2003. Experimental guidance in plant physiology. Beijing: Higher Education Press. [in Chinese] | |
|
赵雨迪, 苏 敏, 陈旭辉. 兰科植物菌根真菌研究概述. 生物学教学, 2023, 48 (11): 2- 4.
doi: 10.3969/j.issn.1004-7549.2023.11.001 |
|
|
Zhao Y D, Su M, Chen X H. Overview of research on mycorrhizal fungi in orchids. Biology Teaching, 2023, 48 (11): 2- 4.
doi: 10.3969/j.issn.1004-7549.2023.11.001 |
|
|
祝丽环, 黄 婷. 种子萌发过程中的生理变化. 生物学教学, 2011, 36 (4): 65- 66.
doi: 10.16590/j.cnki.1001-4705.2025.05.105 |
|
|
Zhu L H, Huang T. Physiological changes during seed germination. Biology Teaching, 2011, 36 (4): 65- 66.
doi: 10.16590/j.cnki.1001-4705.2025.05.105 |
|
| 邹锋康, 王秋红, 周建朝, 等. 生长素调节植物生长发育的研究进展. 中国农学通报, 2018, 34 (24): 34- 40. | |
| Zou F K, Wang Q H, Zhou J C, et al. Auxin regulating plant growth and development: research progress. Chinese Agricultural Science Bulletin, 2018, 34 (24): 34- 40. | |
|
Abuamsha R, Salman M, Ehlers R U. Effect of seed priming with Serratia plymuthica and Pseudomonas chlororaphis to control Leptosphaeria maculans in different oilseed rape cultivars. European Journal of Plant Pathology, 2011, 130 (3): 287- 295.
doi: 10.1007/s10658-011-9753-y |
|
|
Bailly C, El-Maarouf-Bouteau H, Corbineau F. From intracellular signaling networks to cell death: the dual role of reactive oxygen species in seed physiology. Comptes Rendus Biologies, 2008, 331 (10): 806- 814.
doi: 10.1016/j.crvi.2008.07.022 |
|
|
Bischof R H, Ramoni J, Seiboth B. Cellulases and beyond: the first 70 years of the enzyme producer Trichoderma reesei. Microbial Cell Factories, 2016, 15 (1): 106.
doi: 10.1186/s12934-016-0507-6 |
|
|
Dai L, Chen Y W, Wei X L. Hard seed characteristics and seed vigor of Ormosia hosiei. Agriculture, 2023, 13 (5): 1077.
doi: 10.3390/agriculture13051077 |
|
| de Sousa Lopes L, Gallão M I, de Magalhãe Bertini C H C. Mobilisation of reserves during germination of Jatropha seeds. Revista Ciência Agronô mica, 2013, 44 (2): 371- 378. | |
| Dong L, Hao Z, Li Z, et al. Enhancement of welsh onion (Allium fistulosum L.) seed vigor by KNO3 priming. Journal of Agricultural Science and Technology, 2014, 16 (6): 1345- 1353. | |
|
Fernandes B, Dragone G, Abreu A P, et al. Starch determination in Chlorella vulgaris—a comparison between acid and enzymatic methods. Journal of Applied Phycology, 2012, 24 (5): 1203- 1208.
doi: 10.1007/s10811-011-9761-5 |
|
|
Fröhlich V, Feller U. Effect of phloem interruption on endopeptidase and aminopeptidase activities in flag leaves of field-grown wheat. Biochemie und Physiologie der Pflanzen, 1992, 188 (1): 13- 21.
doi: 10.1016/S0015-3796(11)80253-2 |
|
|
Ge M, Wei X L. Spermosphere bacteria promote Ormosia henryi seed germination by activating metabolic pathways. Forests, 2023, 14 (6): 1136.
doi: 10.3390/f14061136 |
|
|
Ge M, Wei X L. Spermosphere bacterial community at different germination stages of Ormosia henryi and its relationship with seed germination. Scientia Horticulturae, 2024, 324, 112608.
doi: 10.1016/j.scienta.2023.112608 |
|
|
Ge M, Wei X L, Fan Y M, et al. The physiological and biochemical mechanisms bioprimed by spermosphere microorganisms on Ormosia henryi seeds. Microorganisms, 2025, 13, 1598.
doi: 10.3390/microorganisms13071598 |
|
|
Lax A R, Vaughn K C. Colocalization of polyphenol oxidase and photosystem II proteins. Plant Physiology, 1991, 96 (1): 26- 31.
doi: 10.1104/pp.96.1.26 |
|
|
Li D X, Ni K K, Zhang Y C, et al. Influence of lactic acid bacteria, cellulase, cellulase-producing Bacillus pumilus and their combinations on alfalfa silage quality. Journal of Integrative Agriculture, 2018, 17 (12): 2768- 2782.
doi: 10.1016/S2095-3119(18)62060-X |
|
|
Lima R B S, de Carvalho Gonçalves J F, Pando S C, et al. Primary metabolite mobilization during germination in rosewood (Aniba rosaeodora Ducke) seeds. Revista Árvore, 2008, 32 (1): 19- 25.
doi: 10.1590/s0100-67622008000100003 |
|
|
Liu R, Lu J, Xing J Y, et al. Transcriptome and metabolome analyses revealing the potential mechanism of seed germination in Polygonatum cyrtonema. Scientific Reports, 2021, 11 (1): 12161.
doi: 10.1038/s41598-021-91598-1 |
|
|
Long X Y, He F J, Sun Y Q, et al. Analysis of main nutritional components in flowers and leaves of 2 Lonicera cuminata germplasm. IOP Conference Series: Earth and Environmental Science, 2019, 237 (5): 052027.
doi: 10.1088/1755-1315/237/5/052027 |
|
| Lutts S, Benincasa P, Wojtyla L, et al. 2016. Seed priming: new comprehensive approaches for an old empirical technique//Araujo S, Balestrazzi A. New challenges in seed biology–basic and translational research driving seed technology. InTech, 1–46. | |
|
Mahadik N D, Puntambekar U S, Bastawde K B, et al. Production of acidic lipase by Aspergillus niger in solid state fermentation. Process Biochemistry, 2002, 38 (5): 715- 721.
doi: 10.1016/S0032-9592(02)00194-2 |
|
|
Mo X, Qian J Y, Liu P, et al. Exogenous betaine enhances the protrusion vigor of rice seeds under heat stress by regulating plant hormone signal transduction and its interaction network. Antioxidants, 2022, 11 (9): 1792.
doi: 10.3390/antiox11091792 |
|
|
Nelson E B. The seed microbiome: origins, interactions, and impacts. Plant and Soil, 2018, 422 (1/2): 7- 34.
doi: 10.1007/s11104-017-3289-7 |
|
|
Nunes I, Hansen V, Bak F, et al. Succession of the wheat seed-associated microbiome as affected by soil fertility level and introduction of Penicillium and Bacillus inoculants in the field. FEMS Microbiology Ecology, 2022, 98 (3): fiac028..
doi: 10.1093/femsec/fiac028 |
|
|
Olofintila O E, Noel Z A. Soybean and cotton spermosphere soil microbiome shows dominance of soilborne copiotrophs. Microbiology Spectrum, 2023, 11, e00377- 23.
doi: 10.1101/2023.01.23.525219 |
|
| Patel Z M, Mahapatra R, Jampala S S M. 2020. Molecular aspects of plant beneficial microbes in agriculture. New York: Academic Press. | |
| Pontes C A, Borges E E D L, Borges R D C G, et al. Seed reserve mobilization of Apuleia leiocarpa (Vogel) J. F. Macbr. (garapa) during imbibition. Revista Á rvore, 2002, 26 (5): 593- 601. | |
| Roslan M A, Zulkifli N N, Sobri Z M, et al. Seed biopriming with P- and K-solubilizing Enterobacter hormaechei sp. improves the early vegetative growth and the P and K uptake of okra (Abelmoschus esculentus) seedling. PLoS One, 2020, 15 (7): e232860. | |
| Saccaram C, Simonin M, Boutet S, et al. Elucidating the interplay between metabolites and microorganisms in the spermosphere of common bean (Phaseolus vulgaris L.) seeds. mSystems, 2025, 10, e00707- 25. | |
| Sahbaz R, Lieberei R, Aniszewski T. Polyphenol oxidase (PPO, catecholase) activity during germination and early seedling growth of Cicer milkvetch (Astragalus cicer L.). Journal of Applied Botany and Food Quality, 2009, 82 (2): 163- 169. | |
| Schiltz S, Gaillard I, Pawlicki-Jullian N, et al. 2015. A review: what is the spermosphere and how can it be studied? Journal of Applied Microbiology, 119(6): 1467–1481. | |
|
Shahzad R, Waqas M, Khan A L, et al. Seed-borne endophytic Bacillus amyloliquefaciens RWL-1 produces gibberellins and regulates endogenous phytohormones of Oryza sativa. Plant Physiology and Biochemistry, 2016, 106, 236- 243.
doi: 10.1016/j.plaphy.2016.05.006 |
|
|
Shen H, He X H, Liu Y Q, et al. A complex inoculant of N2-fixing, P- and K-solubilizing bacteria from a purple soil improves the growth of kiwifruit (Actinidia chinensis) plantlets. Frontiers in Microbiology, 2016, 7, 841.
doi: 10.3389/fmicb.2016.00841 |
|
|
Thipyapong P, Stout M J, Attajarusit J. Functional analysis of polyphenol oxidases by antisense/sense technology. Molecules, 2007, 12 (8): 1569- 1595.
doi: 10.3390/12081569 |
|
|
Tomas-Grau R H, Chalfoun N R, Hael-Conrad V, et al. Induction and suppression of the defense response mediated by two fungal derived molecules in strawberry plants. Acta Horticulturae, 2021, 1309, 781- 788.
doi: 10.17660/actahortic.2021.1309.111 |
|
|
Woo S L, Hermosa R, Lorito M, et al. Trichoderma: a multipurpose, plant-beneficial microorganism for eco-sustainable agriculture. Nature Reviews Microbiology, 2023, 21 (5): 312- 326.
doi: 10.1038/s41579-022-00819-5 |
|
|
Yan R Y, Lin W H, Lu T L, et al. Conjugated hypercrosslinked polymers imprinted with 3, 5-dinitrosalicylic acid for the fluorescent determination of α-amylase activity. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 2023, 291, 122383.
doi: 10.1016/j.saa.2023.122383 |
|
|
Yang R Q, Wang P, Elbaloula M F, et al. Effect of germination on main physiology and biochemistry metabolism of sorghum seeds. Bioscience Journal, 2016, 32 (2): 378- 383.
doi: 10.14393/bj-v32n2a2016-30895 |
|
|
Xu X, Wang C, Xu J, et al. Seed–microbiome interactions: Mechanistic insights and utilization toward seed performance for sustainable agriculture. Plant Communications, 2026, 7 (3): 101716.
doi: 10.1016/j.xplc.2026.101716 |
|
|
Zhao M, Zhang H X, Yan H, et al. Mobilization and role of starch, protein, and fat reserves during seed germination of six wild grassland species. Frontiers in Plant Science, 2018, 9, 234.
doi: 10.3389/fpls.2018.00234 |
|
|
Zhou C C, Xia S Q, Wen Q, et al. Genetic structure of an endangered species Ormosia henryi in southern China, and implications for conservation. BMC Plant Biology, 2023, 23 (1): 220.
doi: 10.1186/s12870-023-04231-w |
|
| Zondo S N N, Mohase L, Tolmay V, et al. Elucidating β-1, 3-glucanase and peroxidase physicochemical properties of wheat cell wall defense mechanism against Diuraphis noxia infestation. Journal of Visualized Experiments, 2024, 209, e66903. |
| [1] | 姜顺邦, 韦小丽. 供水量对花榈木苗期耗水、生长和生理的影响及灌溉制度优化[J]. 林业科学, 2016, 52(10): 22-30. |
| [2] | 徐本美 孙运涛 李锐丽 郭琛 宋宇航. 舞草硬实种子的高活力性状研究[J]. 林业科学, 2006, 42(10): 54-58. |
| [3] | 曹帮华 翟明普 郭俊杰. 不同硬实程度的刺槐种子活力差异性研究[J]. 林业科学, 2005, 41(2): 42-47. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||