

林业科学 ›› 2020, Vol. 56 ›› Issue (12): 166-176.doi: 10.11707/j.1001-7488.20201219
任飞,张佳琦,胡恒康,梁璧,黄有军,娄和强,张启香*
收稿日期:2019-12-03
出版日期:2020-12-25
发布日期:2021-01-22
通讯作者:
张启香
基金资助:Fei Ren,Jiaqi Zhang,Hengkang Hu,Bi Liang,Youjun Huang,Heqiang Lou,Qixiang Zhang*
Received:2019-12-03
Online:2020-12-25
Published:2021-01-22
Contact:
Qixiang Zhang
摘要:
目的: 报告基因是编码易被检测的蛋白质或酶的基因。红色荧光蛋白基因DsRED因其优异的适用特性已作为报告基因广泛应用于动植物以及酵母等真核细胞内基因表达的研究。课题组前期已将DsRED成功转入核桃体细胞胚,阳性体胚生长及增殖正常、阳性再生植株长势良好。本研究通过对核桃体细胞胚和再生植株中DsRED基因的跟踪研究,探讨DsRED作为报告基因在核桃再生植株中的表达是否稳定以及对再生植株的后续生长和发育是否具有影响,同时为进一步拓宽DsRED作为报告基因的应用范围提供参考。方法: 通过荧光显微镜检测核桃体细胞胚、继代培养3年的组培苗以及3年生温室苗的DsRED荧光表达的稳定性,并通过PCR、qRT-PCR以及Western Blot技术检测DsRED基因及其编码蛋白在核桃体细胞胚和组培苗中表达的稳定性。同时,通过对DsRED组培苗及3年生苗的形态指标进行测定,从而明确DsRED作为外源报告基因的可靠性。结果: 核桃DsRED体细胞胚在白光下呈粉红至红色,荧光下呈明亮的红色;与对照相似,DsRED体细胞胚发育也经历了球形胚、心形胚、鱼雷胚和子叶胚阶段,且形态正常。DsRED组培苗和3年生苗在荧光下植株表面呈现明亮的红色,而对照无激发,视野中呈现黑色,且白光下二者表型一致。DsRED组培苗和3年生苗的茎、叶生长参数与对照相比无显著差异。显微结构荧光检测发现,DsRED在组培苗和3年生苗营养器官的维管束部位表达量较高。对核桃DsRED体胚和组培苗进行PCR检测发现DsRED体胚和组培苗均可扩增获得目的条带,与预期条带大小一致(681 bp);qRT-PCR检测结果显示,DsRED组培苗各组织及DsRED体胚的DsRED mRNA表达量无显著性差异,而对照体胚和组培苗中表达量为0。Western Blot检测结果表明,DsRED组培苗和体胚有26 kDa强阳性条带,对照植株中无特异性条带,进一步分析条带灰度,结果表明DsRED组培苗各组织间及DsRED体胚的DsRED表达量无显著性差异,而对照相应结构中表达量均为0。结论: DsRED基因转入核桃体胚后,可在体胚和继代培养3年的组培苗中正常翻译和稳定表达,3年生温室苗DsRED表达稳定且植株表型正常,表明DsRED是核桃理想的遗传转化报告基因。研究结果可为DsRED作为报告基因在果树中的应用提供参考。
中图分类号:
任飞,张佳琦,胡恒康,梁璧,黄有军,娄和强,张启香. 红色荧光蛋白基因DsRED在核桃植株再生过程中的表达稳定性[J]. 林业科学, 2020, 56(12): 166-176.
Fei Ren,Jiaqi Zhang,Hengkang Hu,Bi Liang,Youjun Huang,Heqiang Lou,Qixiang Zhang. Expression Stability of Red Fluorescent Protein Gene DsRED in the Regeneration of Walnut (Juglans regia) Plant[J]. Scientia Silvae Sinicae, 2020, 56(12): 166-176.

图4
红色荧光蛋白基因DsRED在核桃组培苗中稳定表达 A.组培苗1: DsRED组培苗; 2:对照组培苗。B.组培苗外部荧光表达(标尺:3 mm) 1-3:分别为DsRED组培苗白光下根、茎、小叶外形; 4-6:分别为DsRED组培苗荧光下根、茎、小叶外形; 7-9:分别为对照组培苗白光下根、茎、小叶外形; 10-12:分别为对照组培苗荧光下根、茎、小叶外形。C.组培苗显微结构荧光表达(标尺:600 μm) 1-3:分别为DsRED组培苗白光下根、茎、小叶横切面; 4-6:分别为DsRED组培苗荧光下根、茎、小叶横切面; 7-9:对照组培苗白光下根、茎、小叶横切面;10-12:对照组培苗荧光下根、茎、小叶横切面。"

表1
DsRED基因表达对核桃组培苗茎、叶生长的影响①"
| 指标 Index | DsRED (mean±SD) | 对照Control (mean±SD) |
| 株高Plant height/cm | 5.72±0.53 a | 5.69±0.68 a |
| 株幅Plant width/cm | 3.54±0.64 a | 3.43±0.72 a |
| 茎粗Stem diameter/cm | 0.24±0.06 a | 0.26±0.05 a |
| 复叶数Number of compound leaf | 7±2 a | 7±2 a |
| 小叶长Leaflet length/cm | 0.97±0.41 a | 0.98±0.38 a |
| 小叶宽Leaflet width/cm | 0.48±0.35 a | 0.46±0.37 a |
| 叶面积Leaflet area/cm2 | 0.69±0.42 a | 0.74±0.38 a |
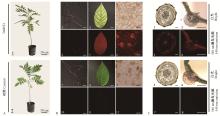
图5
DsRED红色荧光蛋白在3年生苗中稳定表达 A. 3年生苗1:DsRED 3年生苗; 2:对照3年生苗。B. 3年生苗外部荧光表达(标尺3, 6, 9, 12: 10 μm; 其余: 1 cm) 1-3:分别为DsRED 3年生苗根、小叶和叶表皮在白光下;4-6:分别为DsRED 3年生苗根、小叶和叶表皮在荧光下;7-9:分别为对照3年生苗根、小叶和叶表皮在白光下;10-12:分别为对照3年生苗根、小叶和叶表皮在荧光下。C. 3年生苗显微结构荧光表达(标尺:300 μm) 1, 2:分别为DsRED 3年生苗根和小叶在白光下的横切面;3, 4:分别为DsRED 3年生苗根和小叶在荧光下的横切面;5, 6:分别为对照3年生苗根和小叶在白光下的横切面;7, 8:分别为对照3年生苗根和小叶在荧光下的横切面。"
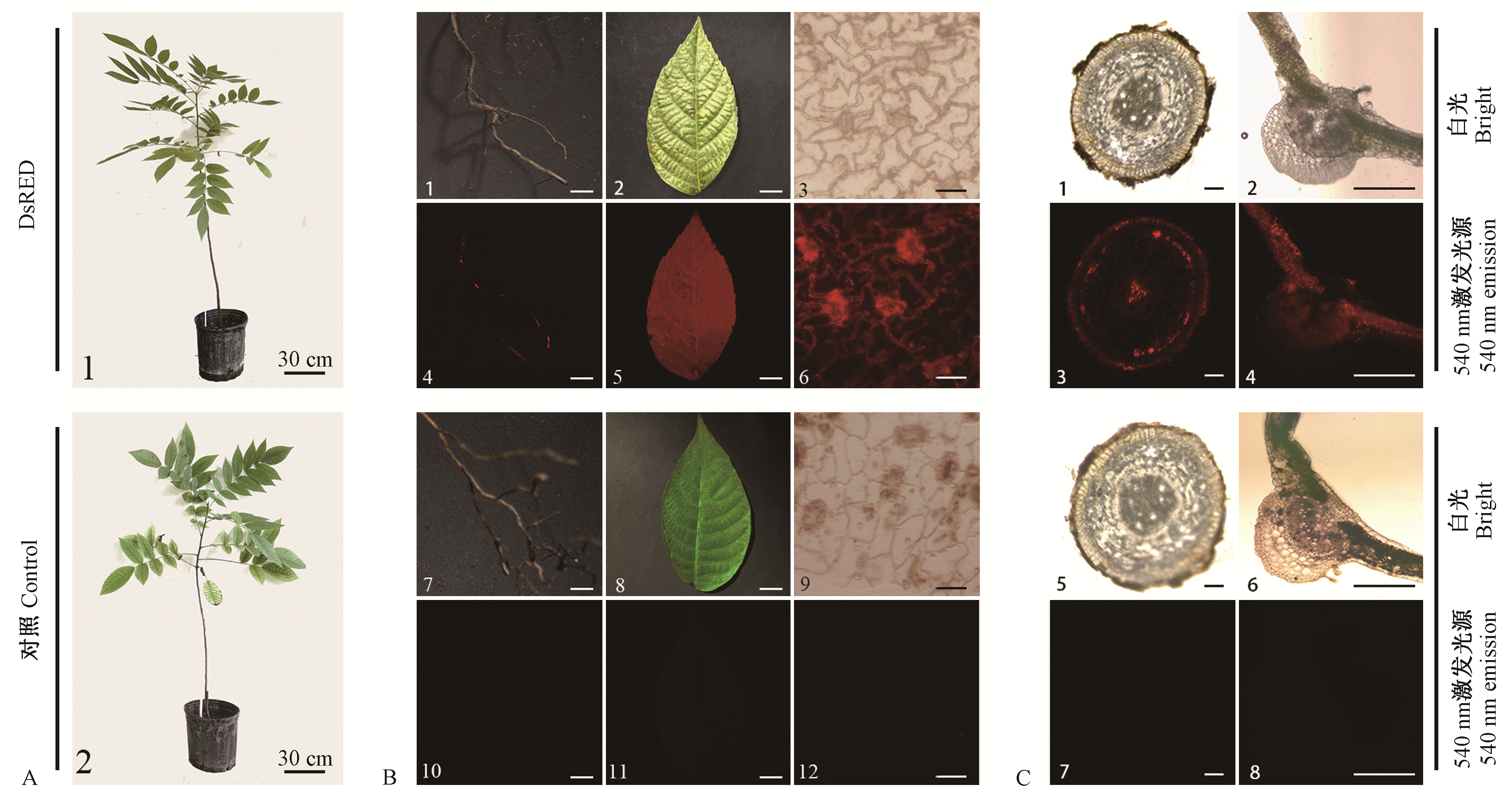
表2
DsRED基因表达对核桃3年生苗茎、叶生长的影响"
| 指标Index | DsRED (mean±SD) | 对照Control (mean±SD) |
| 株高Plant height/cm | 92.88±5.46 a | 94.14±4.37 a |
| 株幅Plant width/cm | 71.78±4.32 a | 69.24±3.75 a |
| 茎粗Stem diameter/cm | 0.89±0.18 a | 0.88±0.19 a |
| 复叶数Number of compound leaf | 11±2 a | 11±2 a |
| 小叶长Leaflet length/cm | 15.32±3.43 a | 14.52±4.53 a |
| 小叶宽Leaflet width/cm | 5.84±1.23 a | 5.63±1.55 a |
| 叶面积Leaflet area/cm2 | 57.41±28.72 a | 50.33±32.19 a |
|
陈茂功, 韩志群, 林小虎, 等. 转DsRed荧光蛋白的新月弯孢Curvularia lunata菌株构建. 植物保护, 2012, 38 (6): 16- 21.
doi: 10.3969/j.issn.0529-1542.2012.06.004 |
|
|
Chen M G , Han Z Q , Lin X H , et al. Construction of DsRed-labeling Curvularia lunata. Plant Protection, 2012, 38 (6): 16- 21.
doi: 10.3969/j.issn.0529-1542.2012.06.004 |
|
| 陈盼飞, 任亚超, 张军, 等. 8年生嫁接转基因杨树Bt毒蛋白的表达与运输. 林业科学, 2016, 52 (7): 46- 52. | |
| Chen P F , Ren Y C , Zhang J , et al. Expression and transportation of Bt toxic protein in 8-year-old grafted transgenic poplar. Scientia Silvae Sinicae, 2016, 52 (7): 46- 52. | |
| 陈盼飞, 左力辉, 王桂英, 等. 盐胁迫下转复合多基因欧美杨107杨幼苗生长及生理响应. 林业科学, 2017, 53 (7): 45- 53. | |
| Chen P F , Zuo L H , Wang G Y , et al. Growth and physiological responses of transgenic Populus×euramericana cv. '4/76' with multiple genes under salt stress. Scientia Silvae Sinicae, 2017, 53 (7): 45- 53. | |
| 高嵩, 何欢, 吕庆雪, 等. 红色荧光蛋白基因DsRED2植物表达载体的构建及遗传转化. 分子植物育种, 2017, 15 (5): 132- 137. | |
| Gao S , He H , Lü Q X , et al. Construction and genetic transformation of DsRED2 plant expression vector in red fluorescent protein gene. Molecular Plant Breeding, 2017, 15 (5): 132- 137. | |
| 李登峰, 杨彩霞, 刘刚. 光声成像报告基因研究进展. 功能与分子医学影像学:电子版, 2018, 7 (4): 41- 45. | |
| Li D F , Yang C X , Liu G . Progress in research on photoacoustic imaging reporter genes. Functional and Molecular Medical Imaging:Electronic Edition, 2018, 7 (4): 41- 45. | |
|
李季, 黄天带, 蔡海滨, 等. 橡胶树转基因植株遗传稳定性分析. 热带作物学报, 2013, 34 (4): 591- 595.
doi: 10.3969/j.issn.1000-2561.2013.04.001 |
|
|
Li J , Huang T D , Cai H B , et al. Genetic stability analyses in transformants of Hevea brasiliensis. Chinese Journal of Tropical Crops, 2013, 34 (4): 591- 595.
doi: 10.3969/j.issn.1000-2561.2013.04.001 |
|
|
李卫东, 王冬梅, 黎小瑛, 等. 用细胞学方法研究番木瓜组培苗的遗传稳定性. 云南植物研究, 2006, 28 (6): 645- 648.
doi: 10.3969/j.issn.2095-0845.2006.06.015 |
|
|
Li W D , Wang D M , Li X Y , et al. Study on the genetic stability of Carica papaya (Caricaceae) propagating plantlets through cytological approach. Acta Botanica Yunnanica, 2006, 28 (6): 645- 648.
doi: 10.3969/j.issn.2095-0845.2006.06.015 |
|
| 李小方, 汤章城, 何玉科. 不定根的形态发生与调节机制. 细胞生物学杂志, 2001, 23 (3): 130- 136. | |
| Li X F , Tang Z C , He Y K . Studies on stability of exogenous gene in transgenic apples. Molecular Plant Breeding, 2001, 23 (3): 130- 136. | |
| 李新锋, 赵淑清. 转基因植物中报道基因GUS的活性检测及其应用. 生命的化学, 2016, 24 (1): 73- 76. | |
| Li X F , Zhao S Q . Assays for GUS activity in transgenic plants and their application in transgenic plants. Chemistry of Life, 2016, 24 (1): 73- 76. | |
|
刘娜, 万瑛, 周镜然, 等. 红色荧光蛋白与卵白蛋白表位融合蛋白的表达与纯化. 免疫学杂志, 2005, 21 (5): 382- 385.
doi: 10.3969/j.issn.1000-8861.2005.05.009 |
|
|
Liu N , Wan Y , Zhou J R , et al. Expression and purification of DsRed tagged OVA epitope fusion protein. Immunological Journal, 2005, 21 (5): 382- 385.
doi: 10.3969/j.issn.1000-8861.2005.05.009 |
|
|
牛东东, 郝育杰, 荣瑞娟, 等. 转基因水稻中GUS蛋白质的检测及其表达特征. 中国农业科学, 2014, 47 (14): 2715- 2722.
doi: 10.3864/j.issn.0578-1752.2014.14.002 |
|
|
Niu D D , Hao Y J , Rong R J , et al. Detection of GUS protein and its expression pattern in transgenic rice plants. Scientia Agricultura Sinica, 2014, 47 (14): 2715- 2722.
doi: 10.3864/j.issn.0578-1752.2014.14.002 |
|
|
汤浩茹, 王永清, 任正隆. 核桃体细胞胚发生与转基因研究进展. 林业科学, 2000, 36 (3): 102- 109.
doi: 10.3321/j.issn:1001-7488.2000.03.017 |
|
|
Tang H R , Wang Y Q , Ren Z L . An overview of progress on somatic embryogenesis and transformation in walnut. Scientia Silvae Sinicae, 2000, 36 (3): 102- 109.
doi: 10.3321/j.issn:1001-7488.2000.03.017 |
|
| 王倩倩, 常腾飞, 师校欣, 等. 外源基因在苹果转基因植株的稳定性研究. 分子植物育种, 2013, 11 (4): 539- 544. | |
| Wang Q Q , Chang T F , Shi X X , et al. Studies on stability of exogenous gene in transgenic apples (Malus domestica Borkh.). Molecular Plant Breeding, 2013, 11 (4): 539- 544. | |
| 王文旭, 郁飞. 红色荧光蛋白融合表达载体的构建和蛋白质细胞内定位研究. 江苏农业科学, 2019, 47 (8): 60- 63. | |
| Wang W X , Yu F . Construction of red fluorescent protein fusion expression vector and intracellular localization of protein. Jiangsu Agricultural Sciences, 2019, 47 (8): 60- 63. | |
|
吴瑞, 张树珍. 绿色荧光蛋白及其在植物分子生物学中的应用. 分子植物育种, 2005, 3 (2): 240- 244.
doi: 10.3969/j.issn.1672-416X.2005.02.014 |
|
|
Wu R , Zhang S Z . Green fluorescent protein and its application in plant molecular biology. Molecular Plant Breeding, 2005, 3 (2): 240- 244.
doi: 10.3969/j.issn.1672-416X.2005.02.014 |
|
|
夏兰芹, 王远, 郭三堆. 外源基因在转基因植物中的表达与稳定性. 生物技术通报, 2000, (3): 8- 12.
doi: 10.3969/j.issn.1002-5464.2000.03.002 |
|
|
Xia L Q , Wang Y , Guo S D . The stability of the expression of foreign genes in transgenic plants. Biotechnology Information, 2000, (3): 8- 12.
doi: 10.3969/j.issn.1002-5464.2000.03.002 |
|
| 杨晓玫, 师尚礼. 红、黄、绿三种颜色荧光质粒导入大肠杆菌中的稳定性表达. 甘肃农业大学学报, 2018, 53 (3): 193- 198. | |
| Yang X M , Shi S L . Gene expression of red, yellow and green fluorescence plasmid stability after transferred in Escherichia coli. Journal of Gansu Agricultural University, 2018, 53 (3): 193- 198. | |
|
杨晓玫, 姚拓, 师尚礼. 荧光蛋白标记研究进展. 草业学报, 2019, 28 (10): 209- 216.
doi: 10.11686/cyxb2019020 |
|
|
Yang X M , Yao T , Shi S L . Progress in fluorescent protein labeling. Acta Prataculturae Sinica, 2019, 28 (10): 209- 216.
doi: 10.11686/cyxb2019020 |
|
| 杨宇, 李江江, 王项, 等. 报告基因及其应用研究进展. 生命科学研究, 2011, 15 (3): 277- 282. | |
| Yang Y , Li J J , Wang X , et al. Progresses on reporter gene and its application. Life Science Research, 2011, 15 (3): 277- 282. | |
|
曾凡锁, 钱晶晶, 康君, 等. 转基因白桦中GUS基因表达的定量分析. 植物学报, 2009, 44 (4): 484- 490.
doi: 10.3969/j.issn.1674-3466.2009.04.010 |
|
|
Zeng F S , Qian J J , Kang J , et al. Histochemical study of β-glucuronidase activity in transgenic birch. Bulletin of Botany, 2009, 44 (4): 484- 490.
doi: 10.3969/j.issn.1674-3466.2009.04.010 |
|
| 曾凡锁, 王晓风, 骆薇, 等. 转基因白桦的花粉活力及外源基因的遗传表达分析. 植物生理学通讯, 2008, 44 (6): 1082- 1086. | |
| Zeng F S , Wang X F , Luo W , et al. Analysis of pollen vigor, inheritance and expression of foreign genes in transgenic birch (Betula platyphylla Suk.) pollen. Plant Physiology Communications, 2008, 44 (6): 1082- 1086. | |
| 赵彦平, 赵春海. 植物转基因育种的分析与研究. 生物技术通报, 2011, 12 (3): 78- 83. | |
| Zhao Y P , Zhao C H . Research and analysis of transgenic breeding in plants. Biotechnology Bulletin, 2011, 12 (3): 78- 83. | |
|
周瑞金, 杜国强, 师校欣. 外源基因在苹果植株中稳定性研究. 东北农业大学学报, 2011, 42 (7): 97- 101.
doi: 10.3969/j.issn.1005-9369.2011.07.018 |
|
|
Zhou R J , Du G Q , Shi X X . Study on stability of exogenous gene in transgenic apple plantlets. Journal of Northeast Agricultural University, 2011, 42 (7): 97- 101.
doi: 10.3969/j.issn.1005-9369.2011.07.018 |
|
| 朱靖杰. 香蕉组织培养中变异的发生与控制途径. 果树科学, 1995, 12 (2): 120- 122. | |
| Zhu J J . The occurrence and control of mutation in banana tissue culture. Journal of Fruit Science, 1995, 12 (2): 120- 122. | |
| Alford S C , Abdelfattah A S , Ding Y , et al. A fluorogenic red fluorescent protein heterodimer. Chemistry & Biology, 2012, 19 (3): 353- 360. | |
|
Czymmek K J , Bourett T M , Sweigard J A , et al. Utility of cytoplasmic fluorescent proteins for live-cell imaging of Magnaporthe grisea in planta. Mycologia, 2002, 94 (2): 280- 289.
doi: 10.1080/15572536.2003.11833234 |
|
|
Dandekar A M , Mcgranahan G H , Vail P V , et al. High levels of expression of full-length cryIA (c) gene from Bacillus thuringiensis in transgenic somatic walnut embryos. Plant Science, 1998, 131 (2): 181- 193.
doi: 10.1016/S0168-9452(97)00256-2 |
|
| Dong Q Y , Liang X F , Zhang W , et al. Fluorescent labeling of Colletotrichum fructicola nuclei based on a reporter gene knock-in strategy. Mycosystema, 2018, 37 (2): 166- 174. | |
|
El-Euch C , Jay-Allemand C , Pastuglia M , et al. Expression of antisense chalcone synthase RNA in transgenic hybrid walnut microcuttings; Effect on flavonoid content and rooting ability. Plant Molecular Biology, 1998, 38 (3): 467- 479.
doi: 10.1023/A:1006034709501 |
|
|
Guo Y , Li W , Sun H , et al. Detection and quantification of Rhizoctonia cerealis in soil using real-time PCR. Journal of General Plant Pathology, 2012, 78 (4): 247- 254.
doi: 10.1007/s10327-012-0390-x |
|
|
Honma Y , Yamakawa T . High expression of GUS activities in sweet potato storage roots by sucrose-inducible minimal promoter. Plant Cell Reports, 2019, 38 (11): 1417- 1426.
doi: 10.1007/s00299-019-02453-7 |
|
| Jach G , Binot E , Frings S , et al. Use of red fluorescent protein from Discosoma sp. (DsRED) as a reporter for plant gene expression. The Plant Journal, 2001, 28 (4): 483- 491. | |
|
Jawed A , Cook J . Reporter genes:Application to the study of mammalian gene transcription. Analytical Biochemistry, 1990, 188 (2): 245- 254.
doi: 10.1016/0003-2697(90)90601-5 |
|
|
Liu X , Walawage S L , Leslie C A , et al. In vitro gene expression and mRNA translocation from transformed walnut (Juglans regia) rootstocks expressing DsRED fluorescent protein to wild-type scions. Plant Cell Reports, 2017, 36 (6): 877- 885.
doi: 10.1007/s00299-017-2116-1 |
|
|
Mcgranahan G H , Leslie C A , Uratsu S L , et al. Improved efficiency of the walnut somatic embryo gene transfer system. Plant Cell Reports, 1990, 8 (9): 512- 516.
doi: 10.1007/BF00820198 |
|
|
Meyer P . Transcriptional transgene silencing and chromatin components. Plant Molecular Biology, 2000, 43 (2/3): 221- 234.
doi: 10.1023/A:1006483428789 |
|
|
Nishizawa K , Kita Y , Kitayama M , et al. A red fluorescent protein, DsRed2, as a visual reporter for transient expression and stable transformation in soybean. Plant Cell Reports, 2006, 25 (12): 1355- 1361.
doi: 10.1007/s00299-006-0210-x |
|
|
Smit P , Raedts J , Portyanko V , et al. NSP1 of the GRAS protein family is essential for rhizobial nod factor-induced transcription. Science, 2005, 308 (5729): 1789- 1791.
doi: 10.1126/science.1111025 |
|
|
Stem M , Mol J , Kooter J . The silencing of genes in transgenic plants. Annals of Botany, 1997, 79 (1): 3- 12.
doi: 10.1006/anbo.1996.0295 |
|
|
Sun L , Alariqi M , Zhu Y , et al. Red fluorescent protein (DsRed2), an ideal reporter for cotton genetic transformation and molecular breeding. The Crop Journal, 2018, 6 (4): 366- 376.
doi: 10.1016/j.cj.2018.05.002 |
|
| Swartz H J . Field performance and phenotypic stability of tissue-culture-propagated strawberries. Journal American Society for Horticultural, 1981, 106, 667- 673. | |
| Taylor C B . Comprehending cosuppression. The Plant Cell, 1997, 9 (8): 1245- 1249. | |
|
Webb C D , Decatur A , Teleman A , et al. Use of green fluorescent protein for visualization of cell-specific gene expression and subcellular protein localization during sporulation in Bacillus subtilis. Journal of Bacteriology, 1995, 177 (20): 5906- 5911.
doi: 10.1128/JB.177.20.5906-5911.1995 |
|
| Wu T M , Lin K C , Liau W S , et al. A set of GFP-based organelle marker lines combined with DsRed-based gateway vectors for subcellular localization study in rice (Oryza sativa L.). Plant Molecular Biology, 2016, 90 (1/2): 107- 115. | |
| Yanushevich Y G , Staroverov D B , Savitsky A P , et al. A strategy for the generation of non-aggregating mutants of Anthozoa fluorescent proteins. Febs Letters, 2002, 511 (1): 11- 14. | |
|
Yarbrough D , Wachter R M , Kallio K , et al. Refined crystal structure of DsRED, a red fluorescent protein from coral, at 2.0-Å resolution. Proceedings of the National Academy of Sciences, 2001, 98 (2): 462- 467.
doi: 10.1073/pnas.98.2.462 |
|
|
Zhang Q , Walawage S L , Tricoli D M , et al. A red fluorescent protein(DsRED) from Discosoma sp. as a reporter for gene expression in walnut somatic embryos. Plant Cell Reports, 2015, 34 (5): 861- 869.
doi: 10.1007/s00299-015-1749-1 |
| [1] | 吴晓雪, 张艾婧, 盖颖, 蒋湘宁. 外源激素对日本落叶松体细胞胚发生不同阶段的影响[J]. 林业科学, 2021, 57(1): 30-39. |
| [2] | 宫峥嵘, 王一峰, 王瀚, 李唯, 耿明建, 张文明, 刘露. 核桃矿质营养研究进展[J]. 林业科学, 2021, 57(1): 178-190. |
| [3] | 王林,代永欣,张劲松,孟平,孙胜,李豪,万贤崇. 水分和光照条件对核桃-黄豆农林复合系统中黄豆光合作用和生长的影响[J]. 林业科学, 2020, 56(4): 188-196. |
| [4] | 梁璧,张佳琦,任飞,胡恒康,徐川梅,胡渊渊,黄有军,娄和强,张启香. 山核桃贝壳杉烯氧化酶基因CcKO的克隆和表达分析[J]. 林业科学, 2020, 56(10): 70-82. |
| [5] | 依巴代提·木合旦尔, 伊丽米努尔, 张毓涛, 刘康. 天山云杉体细胞胚的成熟及萌发条件[J]. 林业科学, 2019, 55(8): 176-183. |
| [6] | 吴丽芳, 魏晓梅, 陆伟东. 白刺花胚性愈伤组织诱导及体细胞胚发生[J]. 林业科学, 2019, 55(7): 170-177. |
| [7] | 周乃富, 宋晓波, 张俊佩, 常英英, 裴东. 核桃芽接愈合的组织学机制[J]. 林业科学, 2019, 55(6): 37-43. |
| [8] | 孙婷玉, 王艳丽, 沈李元, 吴小芹, 朱丽华, 叶建仁. 培养基成分对黑松体胚发育成熟的影响[J]. 林业科学, 2019, 55(4): 178-186. |
| [9] | 陈婷婷, 王鹏凯, 张稼霁, 施季森, 成铁龙, 陈金慧. 脱落酸和玉米素结合使用对杂交鹅掌楸体胚分化及发育的影响[J]. 林业科学, 2019, 55(3): 64-71. |
| [10] | 张佳琦, 胡恒康, 徐川梅, 胡渊渊, 黄有军, 夏国华, 黄坚钦, 常英英, 叶磊, 娄和强, 张启香. 核桃JrGA2ox基因的克隆、亚细胞定位及功能验证[J]. 林业科学, 2019, 55(2): 50-60. |
| [11] | 张韵,刘涛,张涛,谢乐添,黄坚钦,王正加,胡渊渊. 薄壳山核桃果实假果皮的光合特性[J]. 林业科学, 2019, 55(10): 10-18. |
| [12] | 黄仁, 张韵, 张启香, 王正加, 夏国华, 黄坚钦, 胡渊渊. 异源授粉山核桃果皮光合能力差异的转录组分析[J]. 林业科学, 2019, 55(1): 128-137. |
| [13] | 宋跃, 李淑娟, 张含国, 白晓明, 毕显禹, 董实伟, 董昊. 落叶松胚性愈伤组织悬浮培养体系的优化[J]. 林业科学, 2018, 54(7): 146-154. |
| [14] | 曾楚楚, 娄钧翼, 郭明, 王冰璇, 卢闻君. 防治核桃干腐病的新型纳米生物基农药缓释胶囊制备及缓释性能[J]. 林业科学, 2018, 54(5): 87-100. |
| [15] | 周艳威, 陈金慧, 鲁路, 成铁龙, 杨立明, 施季森. 杂交鹅掌楸体胚再生植株淹水胁迫下叶片超微结构及光合特性变化[J]. 林业科学, 2018, 54(3): 19-28. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||