

Scientia Silvae Sinicae ›› 2021, Vol. 57 ›› Issue (8): 33-42.doi: 10.11707/j.1001-7488.20210804
Previous Articles Next Articles
Jinmei Yuan1,2,Jing Luo1,2,Linlin Zhu1,2,Wan Xi1,2,Xumei Zeng1,2,Kangshun Xiong1,2,Caiyun Wang1,2,Riru Zheng1,2,*
Received:2020-05-11
Online:2021-08-25
Published:2021-09-30
Contact:
Riru Zheng
CLC Number:
Jinmei Yuan,Jing Luo,Linlin Zhu,Wan Xi,Xumei Zeng,Kangshun Xiong,Caiyun Wang,Riru Zheng. Free and Glycosylated Aroma Components in Petals of Three Osmanthus fragrans Cultivars[J]. Scientia Silvae Sinicae, 2021, 57(8): 33-42.
Table 1
The volatile components categories and their contents in the full-blossoming period(S3)of Osmanthus fragrans 'Wan Yingui', 'Huangchuan Jingui' and 'Chenghong Dangui'"
| 游离态香气物质 Free aroma compounds | 鉴定方法 Detection methods | 保留指数 Retention index(RI) | 含量Content/(μg·g-1 FW) | ||
| ‘晚银桂’ ‘Wan Yingui’ | ‘潢川金桂’ ‘Huangchuan Jingui’ | ‘橙红丹桂’ ‘Chenghong Dangui’ | |||
| 单萜Monoterpenes | 4.54±1.06 a | 2.19±0.22 b | 0.15±0.01 c | ||
| D-柠檬烯D-Limonene | M, R | 1 018 | 3.42±1.02 a | 2.19±0.22 a | 0.07±0.01 b |
| 顺式-β-罗勒烯Cis-β-Ocimene | M, R, C | 1 030 | 0.12±0.04 a | — | 0.08±0.00 a |
| 单萜醇Monoterpene alcohols | 30.34±7.12 a | 25.84±3.5 ab | 21.92±1.58 b | ||
| 芳樟醇Linalool | M, R, C | 1 094 | 5.48±0.83 a | 4.59±0.28 a | 6.71±0.49 a |
| 2, 6-二甲基-3, 7-辛二烯-2, 6-二醇 3, 7-Octadiene-2, 6-diol, 2, 6-dimethyl | M, R | 1 185 | 3.24±0.55 a | 4.97±0.59 a | — |
| 2, 6-二甲基-1, 7-辛二烯-2, 6-二醇 1, 7-Octadiene-2, 6-diol, 2, 6-dimethyl | M, R | 1 191 | 4.62±0.72 b | 2.51±0.30 b | 6.86±0.50 a |
| 2, 6-二甲基-1, 7-辛二烯-3, 6-二醇 1, 7-Octadiene-2, 6-diol, 3, 6-dimethyl | M, R | 1 256 | 8.93±2.44 b | 13.63±2.20 a | — |
| 3-氧代-α-紫罗醇3-Oxo-α-ionol | M, R | 1 645 | 1.23±0.45 a | 0.09±0.12 b | — |
| 二氢-3-氧代-β-紫罗醇Dihydro-3-oxo-β-ionol | M, R | 1 709 | 6.84±2.13 a | 0.05±0.01 b | 8.35±0.59 a |
| 单萜酮Monoterpene ketones | 135.52±16.89 a | 104.19±13.71 b | 13.08±2.36 c | ||
| 香叶基丙酮Geranyl acetone | M, R | 1 459 | 10.52±2.78 a | 8.91±3.04 a | — |
| α-紫罗酮α-Ionone | M, R, C | 1 423 | 15.74±1.95 a | 6.13±0.28 b | — |
| 二氢-β-紫罗酮Dihydro-β-ionone | M, R, C | 1 434 | 10.41±2.64 a | 3.35±0.03 b | — |
| 反式-β-紫罗酮trans-β-Ionone | M, R, C | 1 483 | 93.44±9.52 a | 84.11±8.67 b | 13.08±2.36 a |
| 4-羟基-β-紫罗酮4-Hydroxy-β-ionone | M, R | 1 664 | 5.41±0.54 a | 1.69±0.22 b | — |
| 单萜氧化物Monoterpene oxides | 24.18±7.53 b | 37.7±2.64 a | 31.02±1.51 a | ||
| 顺式氧化芳樟醇(呋喃型) cis-Linalool oxide(furan) | M, R, C | 1 063 | 0.18±0.04 a | 0.08±0.04 a | 0.13±0.01 a |
| 反式氧化芳樟醇(呋喃型) trans-Linalool oxide(furan) | M, R, C | 1 079 | 0.13±0.05 b | 0.09±0.05 b | 2.43±0.11 a |
| 顺式氧化芳樟醇(吡喃型) cis-Linalool oxide(pyran) | M, R, C | 1 169 | 0.12±0.04 b | 0.32±0.18 | 9.58±0.20 a |
| 反式氧化芳樟醇(吡喃型) trans-Linalool oxide(pyran) | M, R, C | 1 164 | 0.98±0.23 b | 1.20±0.01 b | 5.66±0.12 a |
| 反式8-羟基芳樟醇 trans-8-Hydroxy-linalool | M, R | 1 351 | 14.55±4.76 b | 36.01±2.46 a | 3.57±0.31 c |
| 顺式8-羟基芳樟醇 cis-8-Hydroxy-linalool | M, R | 1 370 | 5.22±2.41 b | — | 9.65±0.67 a |
| 单萜及其衍生物总计 Total monoterpenes and derivatives | 194.58±32.6 b | 254.03±20.17 a | 66.17±5.46 c | ||
Table 2
The glycosylated volatile categories and their contents in the full-blossoming period(S3) of O. fragrans 'Wan Yingui', 'Huangchuan Jingui', and 'ChenghongDangui'"
| 糖苷态香气物质 Glycosylated aroma compounds | 鉴定方法 Detection methods | 保留指数 Retention index(RI) | 含量Content/(μg·g-1 FW) | ||
| ‘晚银桂’ ‘Wan Yingui’ | ‘潢川金桂’ ‘Huangchuan Jingui’ | ‘橙红丹桂’ ‘Cheng hong Dangui’ | |||
| 单萜醇Monoterpene alcohols | 139.04±18.78 a | 99.41±7.07 b | 51.63±5.15 c | ||
| 芳樟醇Linalool | M, R | 1 094 | 3.22±0.43 a | 2.17±0.18 a | 0.50±0.01 b |
| 2, 6-二甲基-3,7 -辛二烯-2, 6-二醇3, 7-Octadiene-2, 6-diol, 2, 6-dimethyl | M, R | 1 185 | 1.23±0.75 b | 3.96±0.26 a | — |
| 3-氧代-α-紫罗醇3-Oxo-α-ionol | M, R | 1 645 | 76.42±9.64 a | 32.58±2.86 b | 25.29±2.76 b |
| 二氢-3-氧代-β-紫罗醇Dihydro-3-oxo-β-ionol | M, R | 1 709 | 56.82±7.42 a | 60.70±3.77 a | 22.06±1.76 b |
| 反式-香叶醇trans-Geraniol | M, R | 1 261 | 1.35±0.54 b | — | 3.78±0.62 a |
| 单萜酮Monoterpene ketones | 226.72±21.5 a | 100.43±6.04 b | 22.62±1.69 c | ||
| 二氢-β-紫罗酮Dihydro-β-ionone | M, R, C | 1 434 | 8.77±1.23 a | 4.01±0.48 b | — |
| 4-羟基-β-紫罗酮4-Hydroxy-β-ionone | M, R | 1 664 | 186.43±13.75 a | 85.41±5.20 b | 7.00±0.89 c |
| 2, 4, 4-三甲基-3-(3-氧代丁基)环己烯-2酮2, 4, 4-Trimethyl-3-(3-oxobutyl)cyclohex-2-enone | M, R | 1 675 | 31.52±6.52 a | 10.15±0.36 b | 15.62±0.80 b |
| 单萜氧化物Monoterpene oxides | 12.34±3.98 b | 13.08±1.34 b | 99.28±3.52 a | ||
| 顺式氧化芳樟醇(呋喃型) cis-Linalool oxide(furan) | M, R, C | 1 063 | 0.83±0.17 a | 1.25±0.08 a | 1.19±0.04 a |
| 反式氧化芳樟醇(呋喃型) trans-Linalool oxide(furan) | M, R, C | 1 079 | 0.45±0.12 b | 0.98±0.06 b | 5.24±0.01 a |
| 反式氧化芳樟醇(吡喃型) trans-Linalool oxide(pyran) | M, R, C | 1 164 | 2.12±0.34 a | 1.50±0.14 a | 1.47±0.30 a |
| 顺式氧化芳樟醇(吡喃型) cis-Linalool oxide(pyran) | M, R, C | 1 169 | — | — | 9.75±1.23 a |
| 反式8-羟基芳樟醇 trans-8-Hydroxy-linalool | M, R | 1 351 | 1.53±0.64 b | 4.17±0.30 b | 25.47±1.08 a |
| 顺式8-羟基芳樟醇 cis-8-Hydroxy-linalool | M, R | 1 370 | 7.41±2.71 b | 5.18±0.76 b | 56.16±0.86 a |
| 单萜及其衍生物总计 Total monoterpenes and derivatives | 378.1±44.26 a | 212.92±14.45 b | 173.53±10.36 b | ||
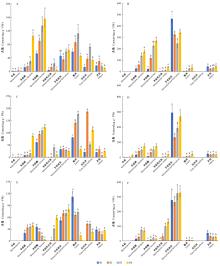
Fig.1
Variation of free and glycosylated aroma compounds in three O. fragrans cultivars at different blossoming periods Free (A) and glycosylated(B) aroma compounds in 'Wan Yingui', free(C) and glycosylated(D) aroma compounds in 'Huangchuan Jingui', free(E) and glycosylated(F) aroma compounds in 'Chenghong Dangui'. S1: The flowers of not blossoming period; S2: Initial blossoming period; S3: Full blossoming period; S4: Late blossoming period. Different small letters indicate significant difference at 0.05 level between the same type of aroma compounds from different blossoming periods. Each sample contains three biological repeats."

|
曹慧, 李祖光, 沈德隆. 桂花品种香气成分的GC-MS指纹图谱研究. 园艺学报, 2009, 36 (3): 391- 398.
doi: 10.3321/j.issn:0513-353X.2009.03.012 |
|
|
Cao H , Li Z G , Shen D L . GC-MS fingerprint analysis of aroma compounds from Osmanthus fragrans Lour. flowers in different varieties. Acta Horticulturae Sinica, 2009, 36 (3): 391- 398.
doi: 10.3321/j.issn:0513-353X.2009.03.012 |
|
|
董尚胜, 童启庆. 栀子花中糖苷酶的分离及其与醇系香气形成的关系. 浙江大学学报: 农业与生命科学版, 2000, 26 (1): 89- 92.
doi: 10.3321/j.issn:1008-9209.2000.01.020 |
|
|
Dong S S , Tong Q Q . Glycosidases separation and their relation to alcohol aroma production in the flowers of Gardenia jasminoides. Journal of Zhejiang University: Agriculture and Life Science, 2000, 26 (1): 89- 92.
doi: 10.3321/j.issn:1008-9209.2000.01.020 |
|
| 侯丹, 付建新, 张超, 等. 桂花品种'堰虹桂'、'玉玲珑'和'杭州黄'的香气成分及释放节律. 浙江农林大学学报, 2015, 32 (2): 208- 220. | |
| Hou D , Fu J X , Zhang C , et al. Flower scent composition of Osmanthus fragrans 'Yanhong Gui', 'Yu Linglong', 'Hangzhou Huang', and their emission patterns. Journal of Zhejiang Agriculture and Forestry University, 2015, 32 (2): 208- 220. | |
|
黄新安, 宛晓春, 夏涛, 等. 茉莉花酶促释香研究. 中山大学学报: 自然科学版, 2009, 48 (2): 144- 148.
doi: 10.3321/j.issn:0529-6579.2009.02.029 |
|
|
Huang X A , Wan X C , Xia T , et al. Investigation on the released aromatic constituents from Jasminum sambac by enzymatic reaction. Acta Scientiarum Naturalium Universitatis Sunyatseni, 2009, 48 (2): 144- 148.
doi: 10.3321/j.issn:0529-6579.2009.02.029 |
|
|
李军, 张淑霞, 郭华强, 等. 紫丁香树皮中羟基苯乙醇类化合物的提取方法及含量研究. 化学与黏合, 2008, 30 (1): 37- 39.
doi: 10.3969/j.issn.1001-0017.2008.01.011 |
|
|
Li J , Zhang S X , Guo H Q , et al. Extraction and determination of hydroxy-phenethyl alcohol compound in bark of Syringa oblata. Chemistry and Adhesion, 2008, 30 (1): 37- 39.
doi: 10.3969/j.issn.1001-0017.2008.01.011 |
|
| 刘偲. 2015. 桂花香气的释放与贮存规律及萜烯合酶OfTPS5的功能分析. 武汉: 华中农业大学硕士学位论文. | |
| Liu C. 2015. The release and storage of floral scent and characterization of OfTPS5 in Osmanthus fragrans Lour. Wuhan: MS thesis of Huazhong Agricultural University. [in Chinese] | |
| 孙美环, 齐欣, 唐向阳, 等. 由月桂烯合成香叶基丙酮的研究. 化学工业与工程, 2016, 33 (1): 51- 56. | |
| Sun M H , Qi X , Tang X Y , et al. Synthesis of geranyl acetone from myrcene. Chemical Industry and Engineering, 2016, 33 (1): 51- 56. | |
| 田怀香, 李凤华, 吴艳. 利用电子鼻分析不同品种的桂花香气. 林业科技开发, 2014, 28 (4): 93- 96. | |
| Tian H X , Li F H , Wu Y . Fragrance identification for three cultivars in Osmanthus fragrans by electronic nose. China Forestry Science and Technology, 2014, 28 (4): 93- 96. | |
| 向其柏, 刘玉莲. 2008. 中国桂花品种图志. 杭州: 浙江科学技术出版社, 2-88. | |
| Xiang Q B, Liu Y L. 2008. An illustrated monograph of the sweet osmanthus cultivars in China. Hangzhou: Zhejiang Science and Techonology Press, 2-88. [in Chinese] | |
|
杨志萍, 姚卫蓉, 钱和. β-D-葡萄糖苷酶对桂花香气成分的影响. 精细化工, 2005, 22 (12): 924- 926.
doi: 10.3321/j.issn:1003-5214.2005.12.012 |
|
|
Yang Z P , Yao W R , Qian H . Effect of β-D-glucosidase on aromatic compounds of sweet osmanthus. Fine Chemicals, 2005, 22 (12): 924- 926.
doi: 10.3321/j.issn:1003-5214.2005.12.012 |
|
|
姚卫蓉, 陈军杰, 钱和. 玫瑰花中键合态和游离态组分的分析. 食品科学, 2007, 28 (11): 487- 492.
doi: 10.3321/j.issn:1002-6630.2007.11.116 |
|
|
Yao W R , Chen J J , Qian H . Analysis of free and glycosylated aroma components of rose. Food Science, 2007, 28 (11): 487- 492.
doi: 10.3321/j.issn:1002-6630.2007.11.116 |
|
| 钟娴. 2014. '平潭水仙'β-葡萄糖苷酶与挥发性芳香物质形成关系的研究. 福州: 福建农林大学硕士学位论文. | |
| Zhong X. 2014. The study of relationship of β-glucosidase and volatile aromatic components of Narcissus tazetta L. var. chinensis Roem. Fuzhou: MS thesis of Fujian Agriculture and Forestry University. [in Chinese] | |
| 邹晶晶, 蔡璇, 曾祥玲, 等. 桂花不同品种开花过程中香气活性物质的变化. 园艺学报, 2017, 44 (8): 1517- 1534. | |
| Zou J J , Cai X , Zeng X L , et al. Changes of aroma-active compounds in different cultivars of Osmanthus fragrans during flowering. Acta Horticulturae Sinica, 2017, 44 (8): 1517- 1534. | |
|
Baldermann S , Kato M , Kurosawa M , et al. Functional characterization of a carotenoid cleavage dioxygenase 1 and its relation to the carotenoid accumulation and volatile emission during the floral development of Osmanthus fragrans Lour. Journal of Experimental Botany, 2010, 61 (11): 2967- 2977.
doi: 10.1093/jxb/erq123 |
|
| Boachon B , Junker R R , Miesch L , et al. CYP76C1(Cytochrome P450)-mediated linalool metabolism and the formation of volatile and soluble linalool oxides in Arabidopsis flowers: a strategy for defense against floral antagonists. Plant Cell, 2015, 27 (10): 2972- 2990. | |
|
Bönisch F , Frotscher J , Stanitzek S , et al. Activity-based profiling of a physiologic aglycone library reveals sugar acceptor promiscuity of family UDP-glucosyltransferases from grape. Plant Physiology, 2014, 166 (1): 23- 39.
doi: 10.1104/pp.114.242578 |
|
| Cai X , Mai R Z , Zou J J , et al. Analysis of aroma-active compounds in three sweet osmanthus(Osmanthus fragrans) cultivars by GC-olfactometry and GC-MS. Journal of Zhejiang University-SCIENCE . Biomedicine and Biotechnology, 2014, 15 (7): 638- 648. | |
|
D'Onofrio C , Matarese F , Cuzzola A . Study of the terpene profile at harvest and during berry development of Vitis vinifera L. aromatic varieties Aleatico, Brachetto, Malvasia di Candia aromatica and Moscato bianco. Journal of the Science of Food and Agriculture, 2017, 97 (9): 2898- 2907.
doi: 10.1002/jsfa.8126 |
|
|
Dudareva N , Klempien A , Muhlemann J K , et al. Biosynthesis, function and metabolic engineering of plant volatile organic compounds. New Phytologist, 2013, 198 (1): 16- 32.
doi: 10.1111/nph.12145 |
|
|
Friedericke B , Johanna F , Sarah S , et al. A UDP-glucose: monoterpenol glucosyltransferase adds to the chemical diversity of the grapevine metabolome. Plant Physiology, 2014a, 165 (2): 561- 581.
doi: 10.1104/pp.113.232470 |
|
|
Friedericke B , Johanna F , Sarah S , et al. Activity-based profiling of a physiologic aglycone library reveals sugar acceptor promiscuity of family 1 UDP-glucosyltransferases from grape. Plant Physiology, 2014b, 166 (1): 23- 39.
doi: 10.1104/pp.114.242578 |
|
|
Fu J , Hou D , Wang Y G , et al. Identification of floral aromatic volatile compounds in 29 cultivars from four groups of Osmanthus fragrans by gas chromatography-mass spectrometry. Horticulture, Environment and Biotechnology, 2019, 60 (5): 611- 623.
doi: 10.1007/s13580-019-00153-5 |
|
|
Fu J X , Hou D , Zhang C , et al. The emission of the floral scent of four Osmanthus fragrans cultivars in response to different temperatures. Molecules, 2017, 22 (3): 430.
doi: 10.3390/molecules22030430 |
|
|
Green S A , Chen X , Nieuwenhuizen N J , et al. Identification, functional characterization, and regulation of the enzyme responsible for floral(E)-nerolidol biosynthesis in kiwifruit(Actinidia chinensis). Journal of Experimental Botany, 2012, 63 (5): 1951- 1967.
doi: 10.1093/jxb/err393 |
|
|
Han Y J , Wang H Y , Wang X D , et al. Mechanism of floral scent production in Osmanthus fragrans and the production and regulation of its key floral constituents, β-ionone and linalool. Horticulture Research, 2019, 6 (1): 106.
doi: 10.1038/s41438-019-0189-4 |
|
|
He Y X , Yuan W J , Dong M F , et al. The first genetic map in sweet osmanthus(Osmanthus fragrans Lour.) using specific locus amplified fragment sequencing. Frontiers in Plant Science, 2017, 8, 1621.
doi: 10.3389/fpls.2017.01621 |
|
|
Junker R R , Gershenzon J , Unsicker S B . Floral odor bouquet loses its ant repellent properties after inhibition of terpene biosynthesis. Journal of Chemical Ecology, 2011, 37 (12): 1323- 1331.
doi: 10.1007/s10886-011-0043-0 |
|
|
Lewinsohn E , Schalechet F , Wilkinson J , et al. Enhanced levels of the aroma and flavor compound S-linalool by metabolic engineering of the terpenoid pathway in tomato fruits. Plant Physiology, 2001, 127 (3): 1256- 1265.
doi: 10.1104/pp.010293 |
|
|
Lucker J , Bouwmeester H J , Schwab W , et al. Expression of Clarkia S-linalool synthase in transgenic petunia plants results in the accumulation of S-linalyl-β-d-glucopyranoside. The Plant Journal, 2001, 27 (4): 315- 324.
doi: 10.1046/j.1365-313x.2001.01097.x |
|
|
Molly C H , Santiago R R . Evolution and diversity of floral scent chemistry in the euglossine bee-pollinated orchid genus Gongora. Annals of Botany, 2016, 118 (1): 135- 148.
doi: 10.1093/aob/mcw072 |
|
|
Parker M , Capone D L , Francis I L , et al. Aroma precursors in grapes and wine: flavor release during wine production and consumption. Journal of Agricultural and Food Chemistry, 2018, 66 (10): 2281- 2286.
doi: 10.1021/acs.jafc.6b05255 |
|
| Picone J M , Clery R A , Watanabe N , et al. Rhythmic emission of floral volatiles from Rosa damascena semperflorens cv. 'Quatre Saisons'. Planta, 2004, 219 (3): 468- 478. | |
|
Raguso R A . More lessons from linalool: insights gained from a ubiquitous floral volatile. Current Opinion in Plant Biology, 2016, 32, 31- 36.
doi: 10.1016/j.pbi.2016.05.007 |
|
| Shoji O , Eichiro O , Manabu H , et al. Volatile glycosylation in tea plants: sequential glycosylation for the biosynthesis of aroma β-primeverosidesare catalyzed by two Camellia sinensis glycosyltransferases. Plant Physiology, 2015, 168 (2): 467- 477. | |
|
Simona B , Christian G , Mathlde C , et al. Changes in volatiles and glycosides during fruit maturation of two contrasted tomato(Solanum lycopersicum) lines. Journal of Agricultural and Food Chemistry, 2009, 57 (2): 591- 598.
doi: 10.1021/jf8023062 |
|
|
Song C K , Härtl K , McGraphery K , et al. Attractive but toxic: emerging roles of glycosidically bound volatiles and glycosyltransferases involved in their formation. Molecular Plant, 2018, 11 (10): 1225- 1236.
doi: 10.1016/j.molp.2018.09.001 |
|
|
Wang L M , Li M T , Jin W W , et al. Variations in the components of Osmanthus fragrans Lour. essential oil at different stages of flowering. Food Chemistry, 2009, 114 (1): 233- 236.
doi: 10.1016/j.foodchem.2008.09.044 |
|
|
Williams P J , Strauss C R , Wilson B . Hydroxylated linalool derivatives as precursors of volatile monoterpenes of muscat grapes. Journal of Agricultural and Food Chemistry, 1980, 28 (4): 766- 771.
doi: 10.1021/jf60230a037 |
|
| Wu B P , Cao X M , Liu H R , et al. A UDP-glucosyltransferase PpUGT85A2 controls volatile glycosylation in peach. Journal of Experimental Botany, 2018, 70 (30): 925- 936. | |
|
Xin H , Wu B , Zhang H , et al. Characterization of volatile compounds in flowers from four groups of sweet osmanthus(Osmanthus fragrans) cultivars. Canadian Journal of Plant Science, 2013, 93 (5): 923- 931.
doi: 10.4141/cjps2012-333 |
|
|
Yar-Khing Y , Claire G , Mindy Y W , et al. Manipulation of flavour and aroma compound sequestration and release using a glycosyltransferase with specificity for terpene alcohols. Plant Journal, 2014, 80 (2): 317- 330.
doi: 10.1111/tpj.12634 |
|
| Zeng X L , Liu C , Zheng R R , et al. Emission and accumulation of monoterpene and the key terpene synthase(TPS) associated with monoterpene biosynthesis in Osmanthus fragrans Lour. Frontiers in Plant Science, 2015, 6, 1232. | |
|
Zheng R R , Liu C , Wang Y L , et al. Expression of MEP pathway genes and non-volatile sequestration are associated with circadian rhythm of dominant terpenoids emission in Osmanthus fragrans Lour. Frontiers in Plant Science, 2017, 8, 1869.
doi: 10.3389/fpls.2017.01869 |
| [1] | Wang Jun, Wang Yuguang, Yang Jinling, Li Wei, Dong Wenhua, Mei Wenli, Dai Haofu. Comparison of the Anatomy Structure and Chemical Compositions of Agarwoods from Two Kinds of Aquilaria sinensis [J]. Scientia Silvae Sinicae, 2019, 55(7): 146-154. |
| [2] | Qiu Shuai, Shen Baichun, Li Tingting, Guo Juan, Wang Ji, Sun Lina, Chen Xuping, Hu Shaoqing. A Method of Osmanthus fragrans Cultivars Identification Based on Random Forest Algorithm and SRAP Molecular Markers [J]. Scientia Silvae Sinicae, 2018, 54(1): 32-45. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||